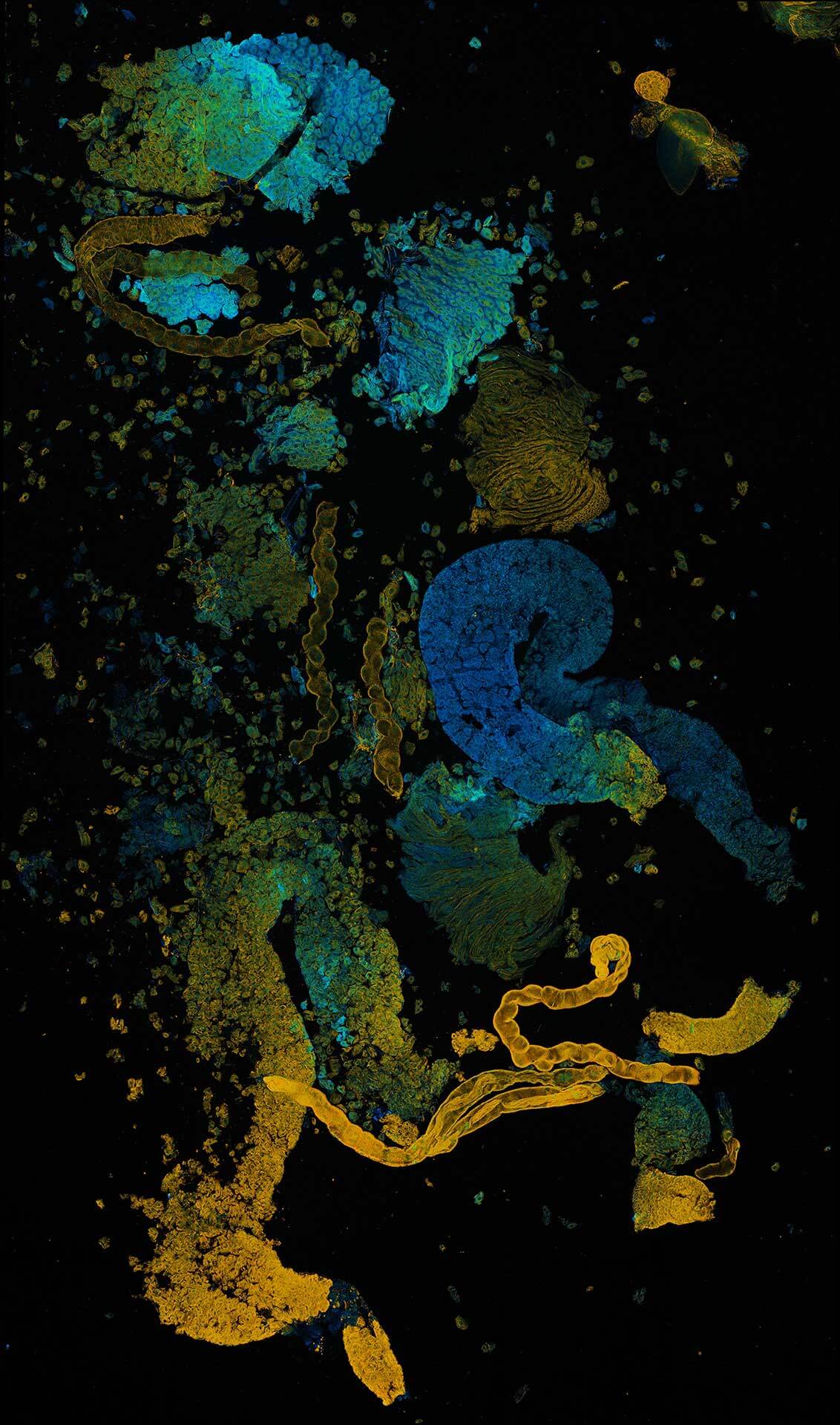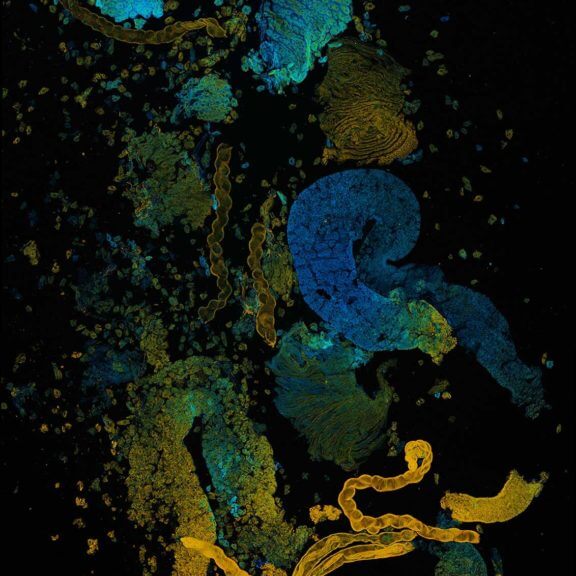Sample gallery
Fluorescence imaging, whether at confocal, STED or MINFLUX resolution, guarantees unique insights into the function and structure of life at the molecular level. Besides the scientific information content, some sample portraits provide simply beautiful images. Enjoy browsing our sample gallery.
the fine art of science
Description
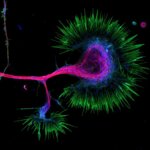
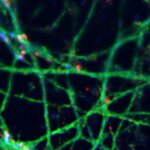
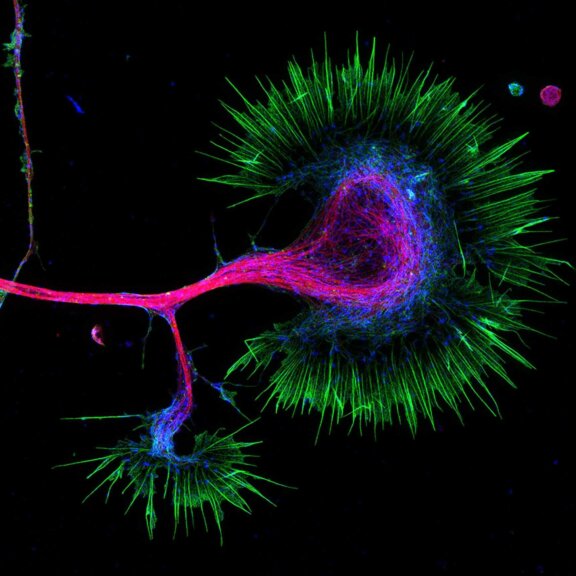
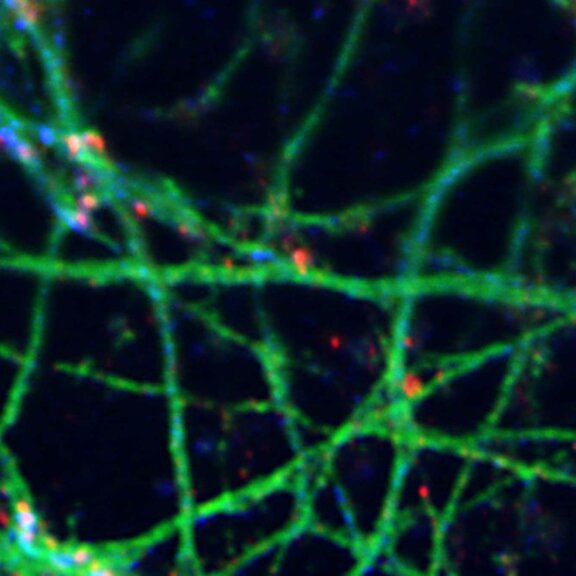
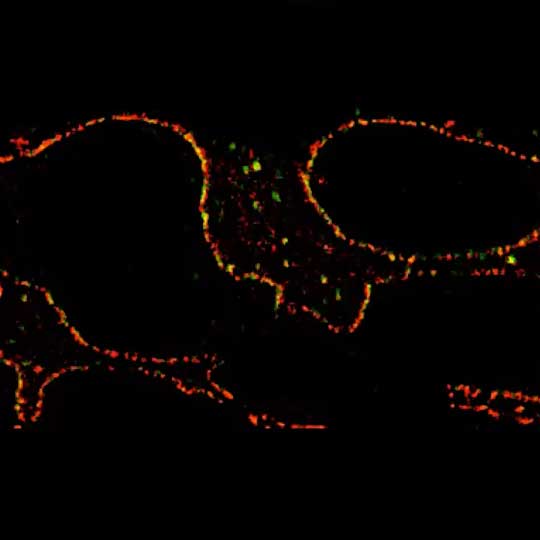
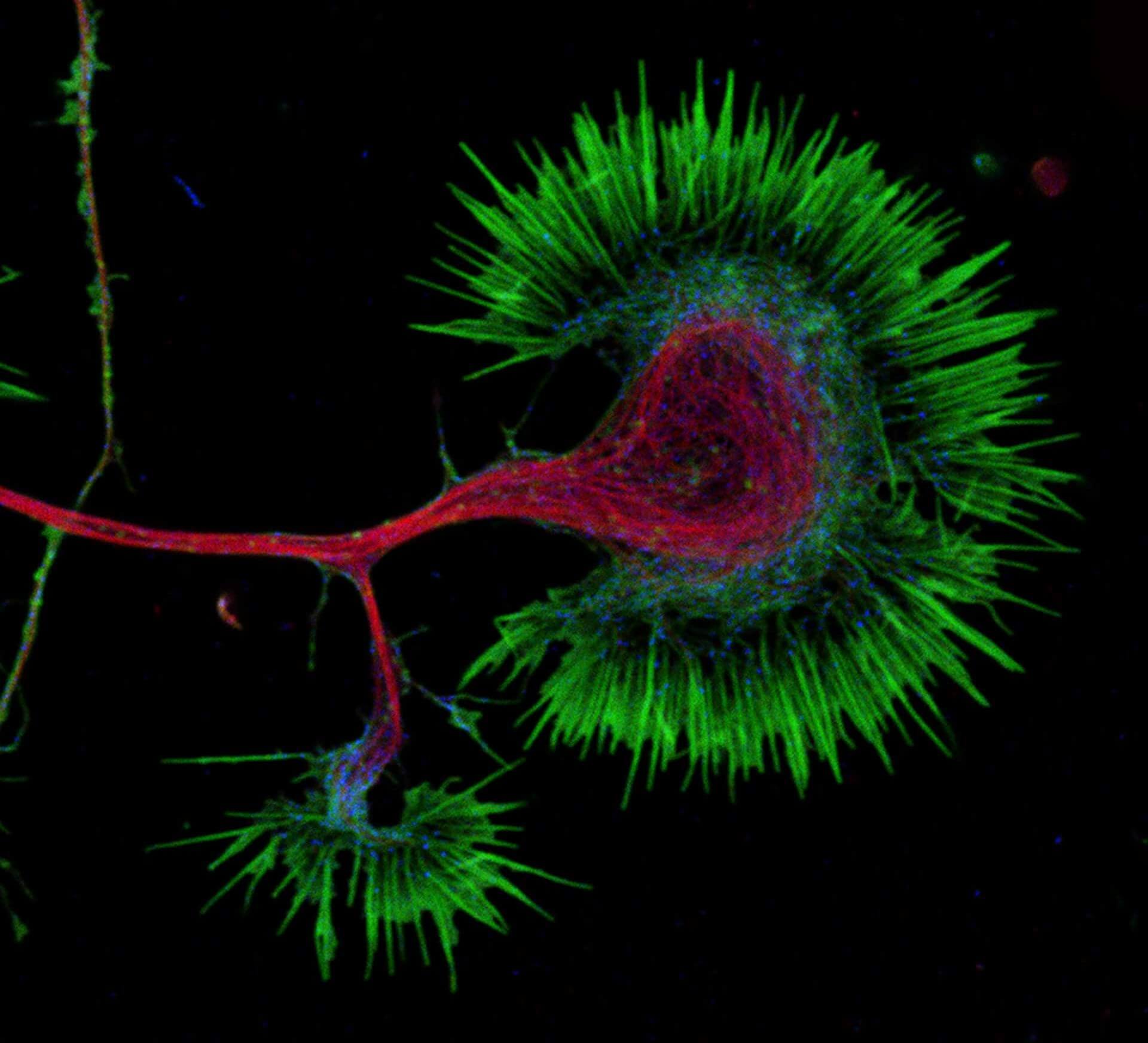
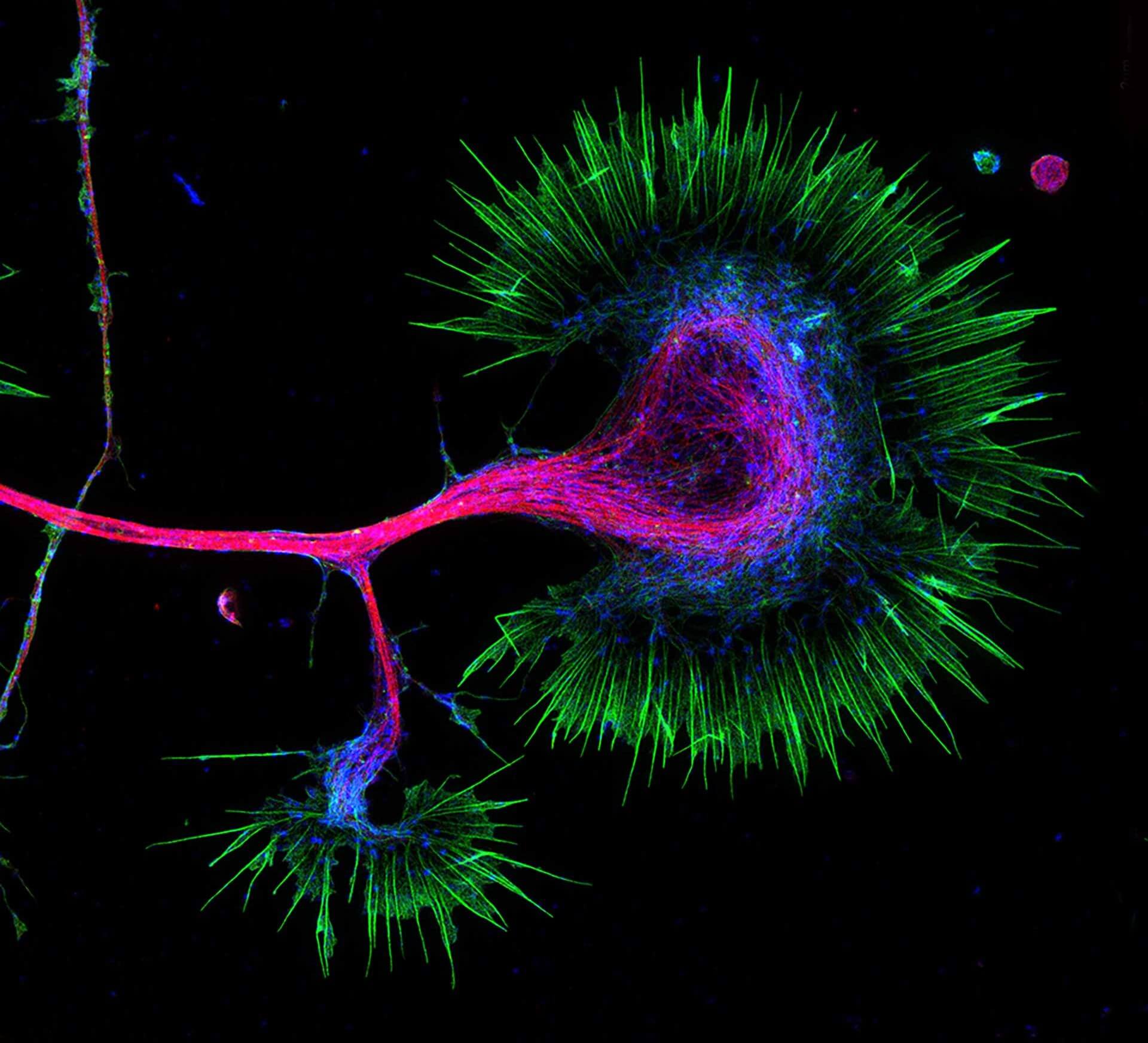
Growth cone at the tip of the axon of a primary hippocampal neuron at 1 day in vitro. Microtubules (Tuj1, abberior STAR 580, red) are bundled in the central-domain suggesting a pausing state. The molecular motor myosin IIB (confocal, Alexa488, blue) is enriched at the transition-zone, along the F-actin arcs. In the peripheral domain actin forms bundles in the filopodia (Phalloidin, abberior STAR 635, green). Sample courtesy: Elisa D'Este, Max Planck Institute for Biophysical Chemistry, Göttingen, Germany.
Description
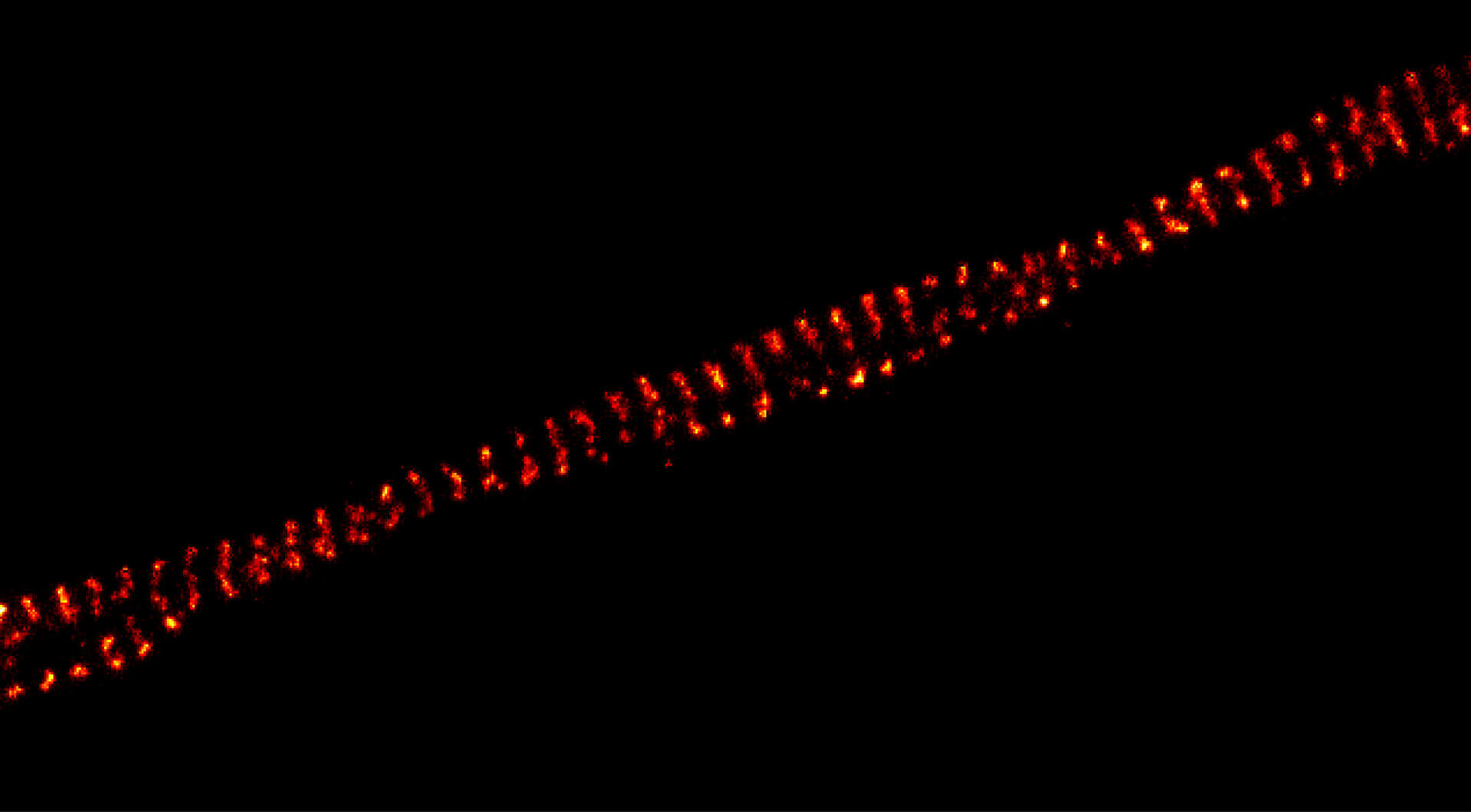
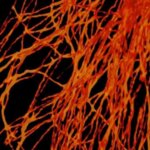
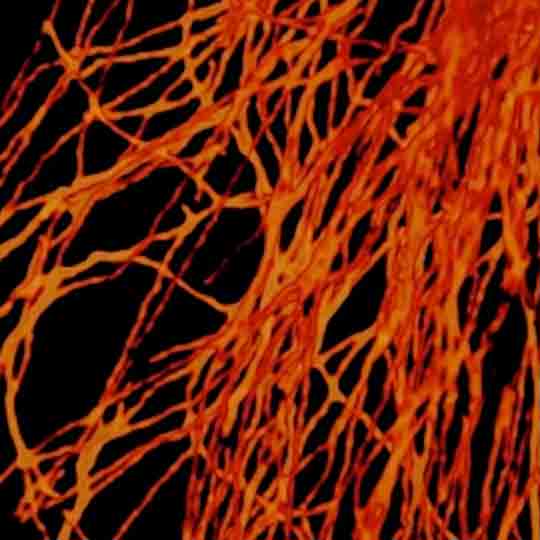
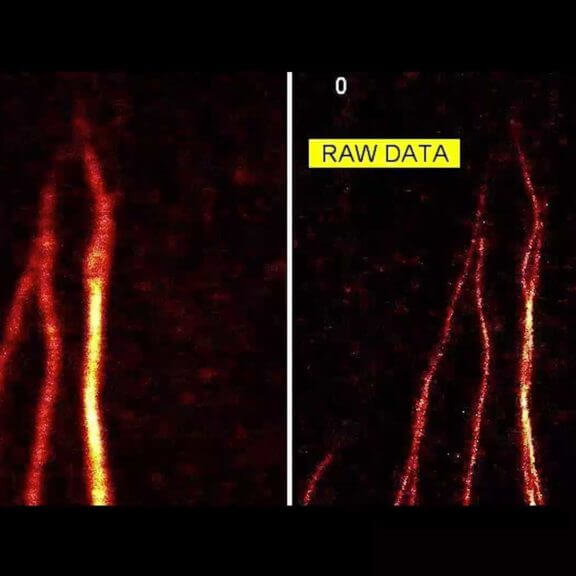
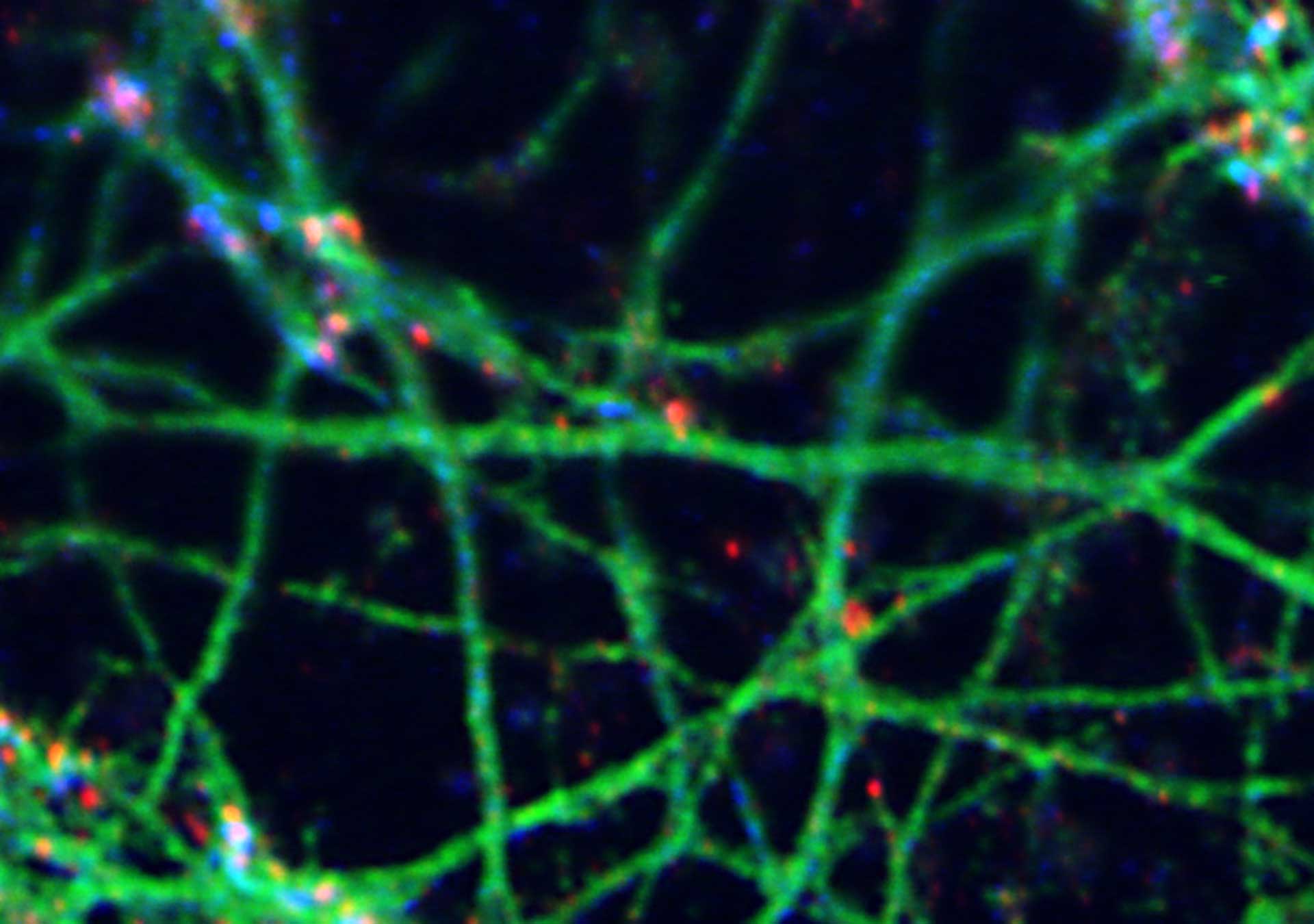
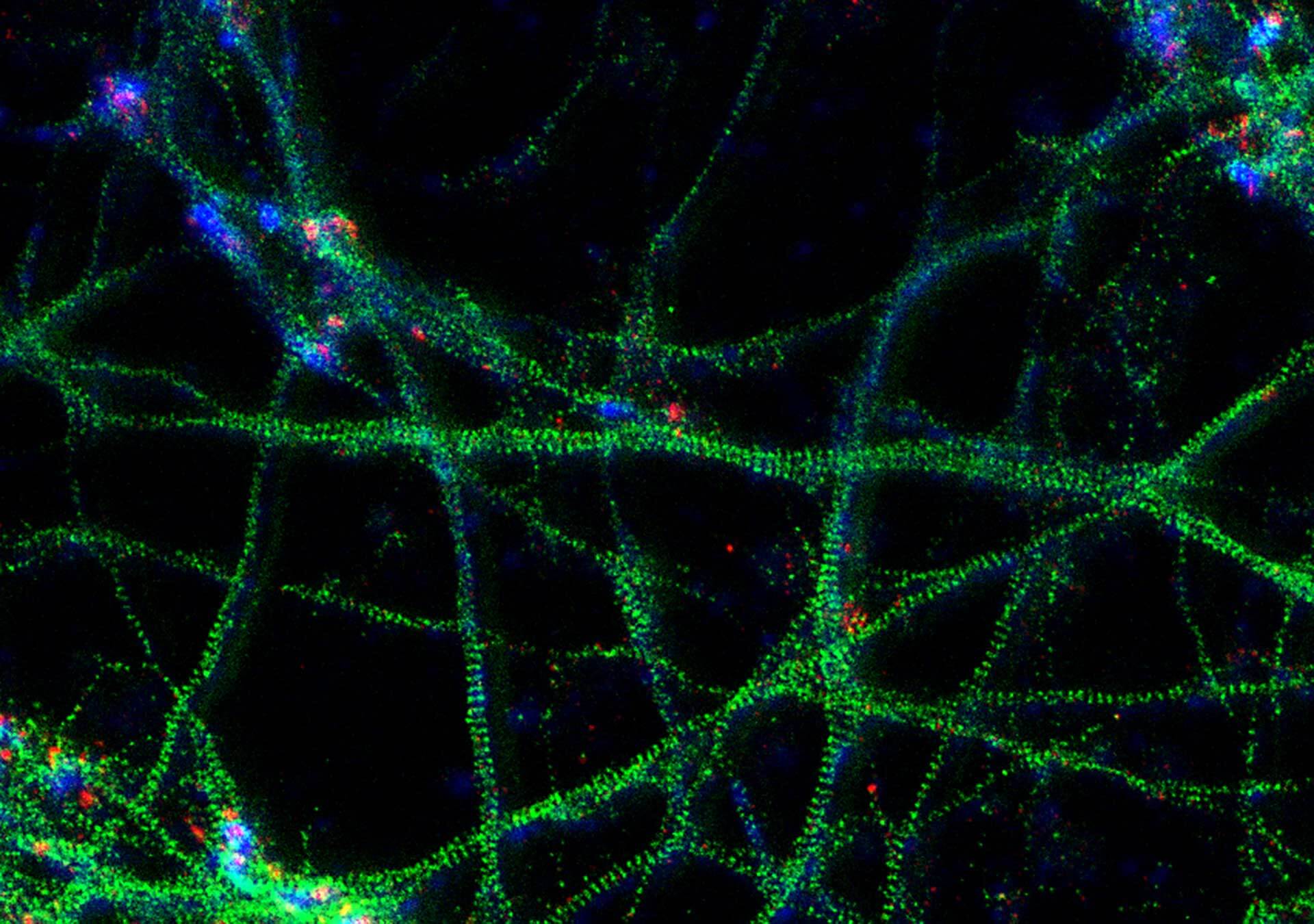
3-color STED image of primary hippocampal neurons. Please note the characteristic ~190 nm beta II spectrin periodicity along distal axons (green) which is only visible in the STED image. Labelled structures: beta II spectrin (green, abberior STAR 635P), Bassoon (red, abberior STAR 580), Actin cytoskeleton (blue, phalloidin, Oregon Green 488). Imaged with abberior Expert Line with 595nm and 775nm STED laser. Sample was prepared by Elisa D’Este @ MPIBPC, Göttingen.
Modules:
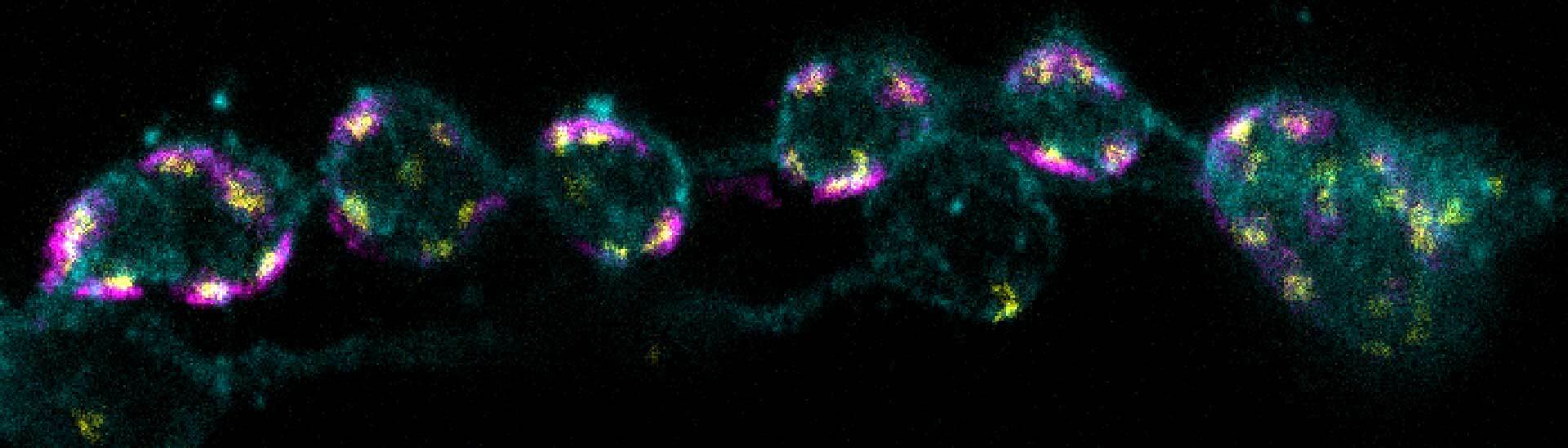
Description
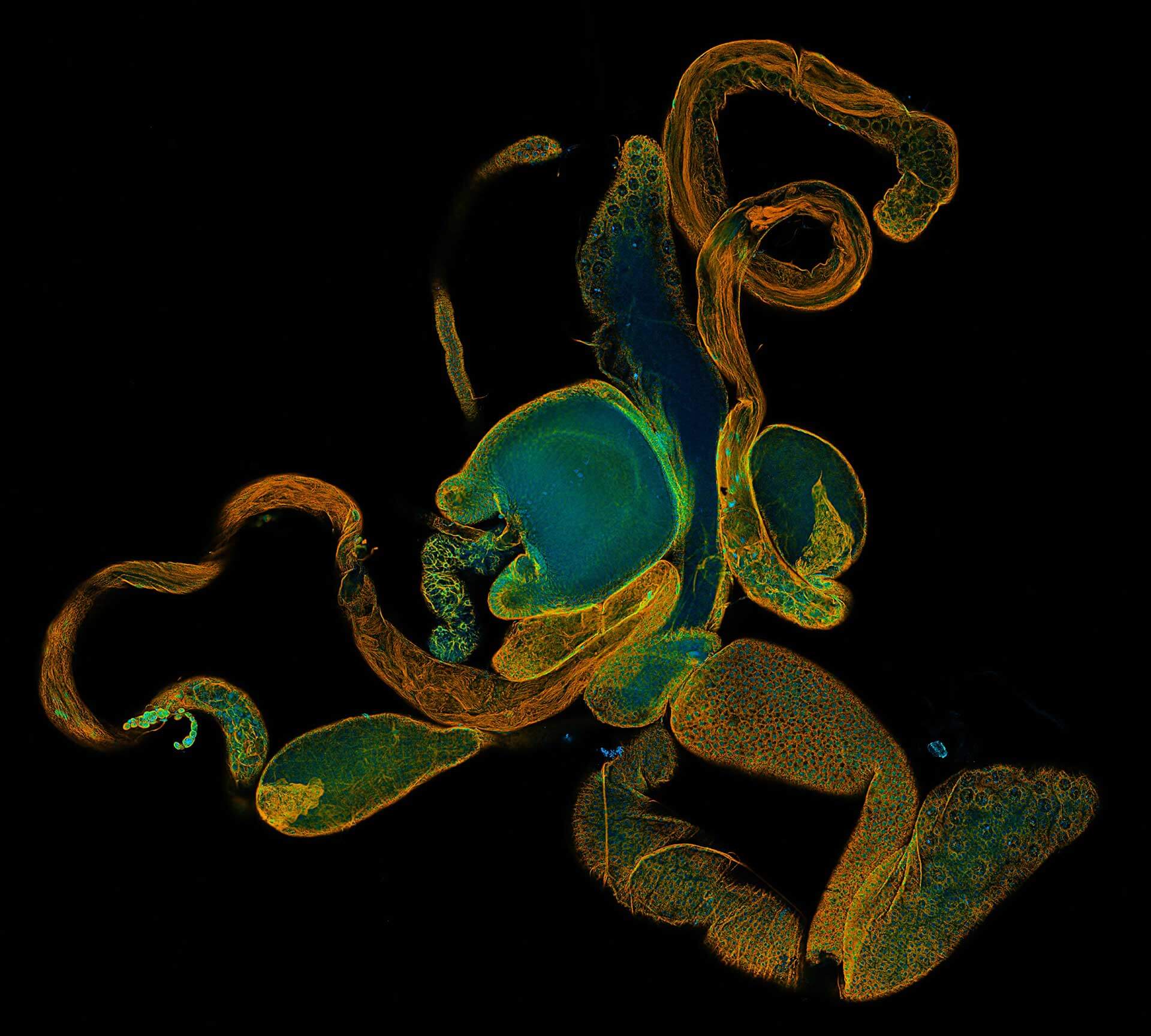
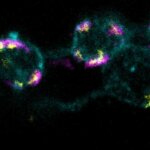
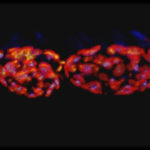
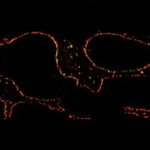
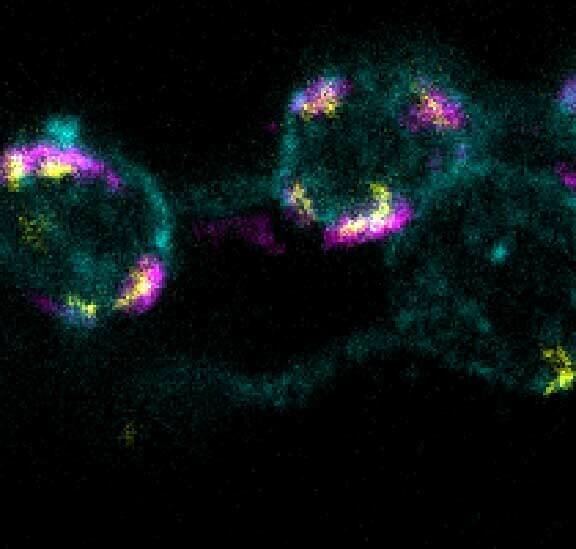
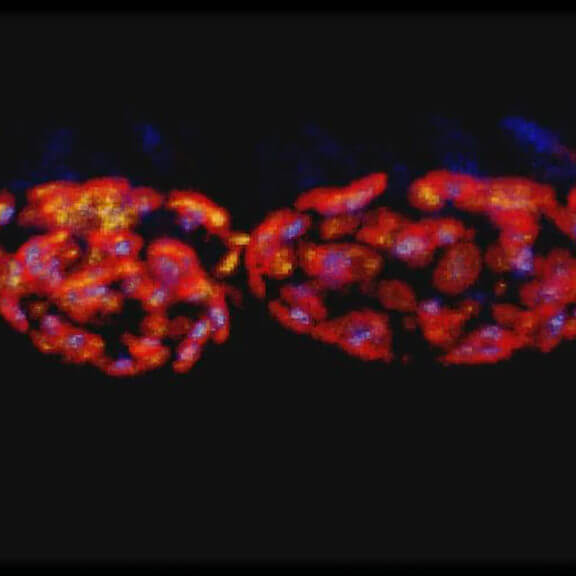
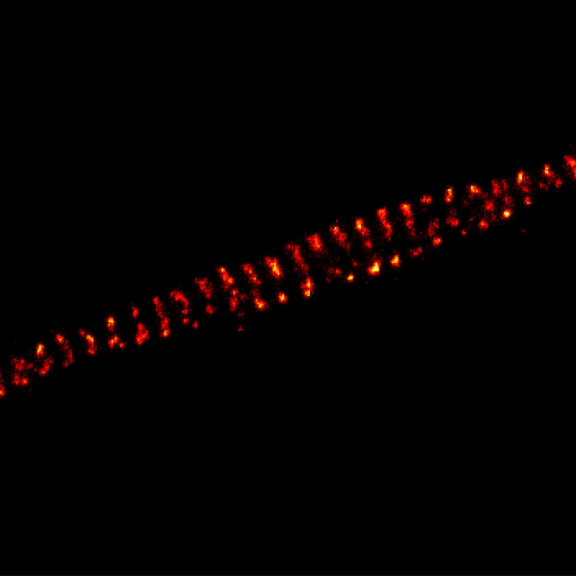
3-color STED imaging: active zones at the Drosophila larval neuromuscular junction immunostained for Bruchpilot and two other proteins.
Two superresolution channels (magenta, yellow) using a 775nm STED laser & one superresolution channel using a 595nm STED laser.
Samples by M. Lenz & M. Landgraf (University of Cambridge, UK).
Description
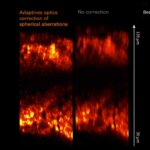
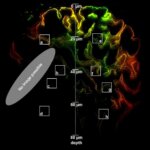
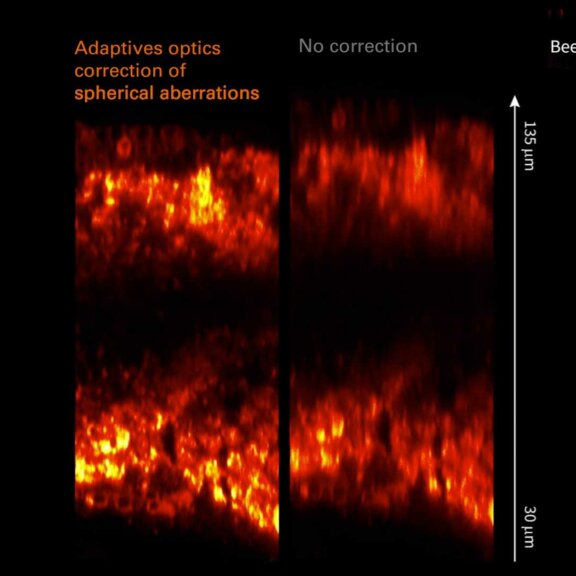
Spherical aberrations arising from index mismatch between sample and immersion are corrected for, as well as higher-order, sample-induced aberrations.
Without Adaptive Optics, excitation laser power is typically increased with focus depth in order to compensate loss of signal due to aberrations. Adaptive Optics preserves resolution and brightness deep inside thick samples and enables imaging at low light levels.
Sample courtesy of Amelie Cabirol and Albrecht Haase, University of Trento.
Modules:
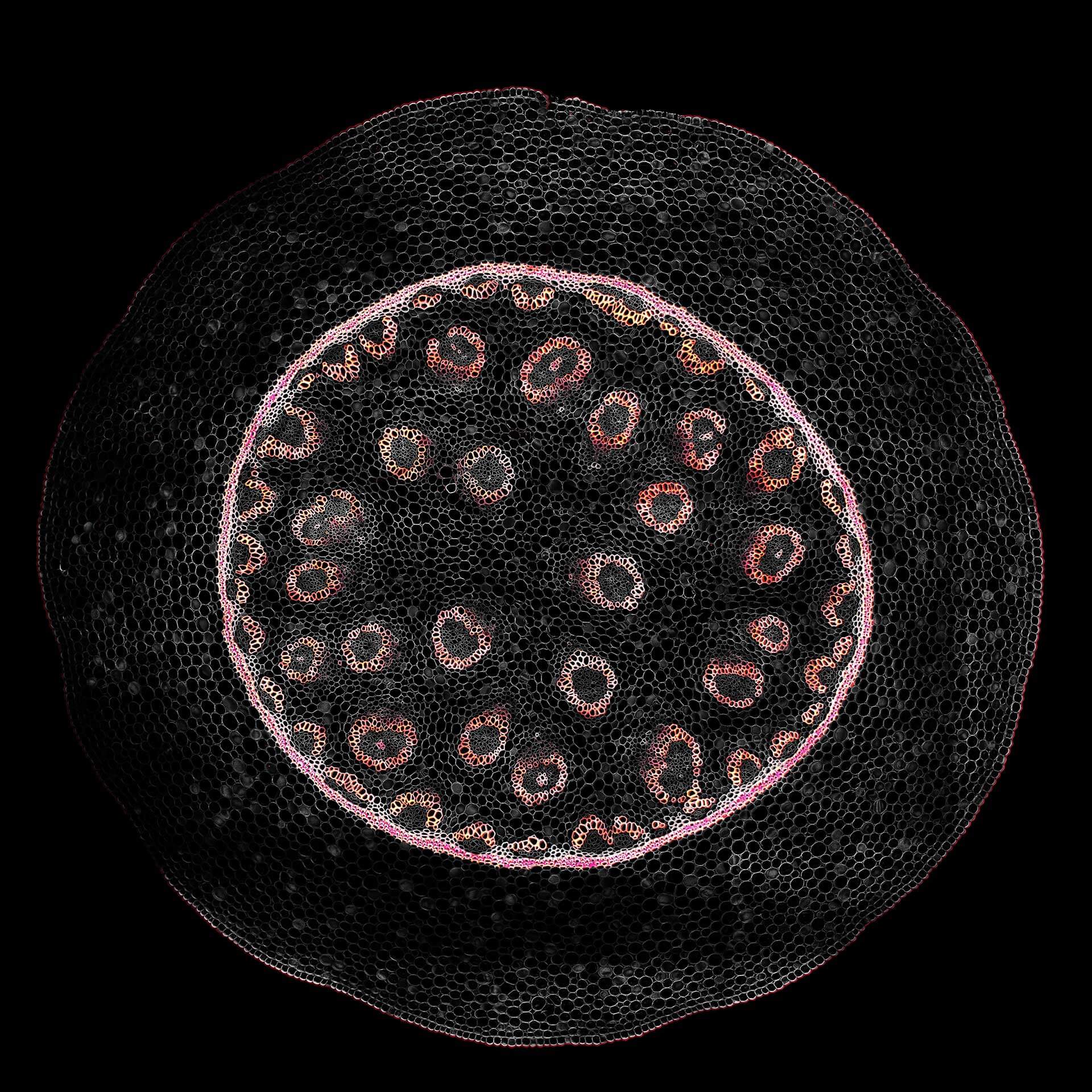
Description
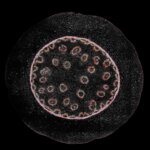
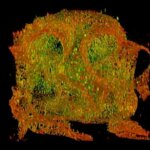
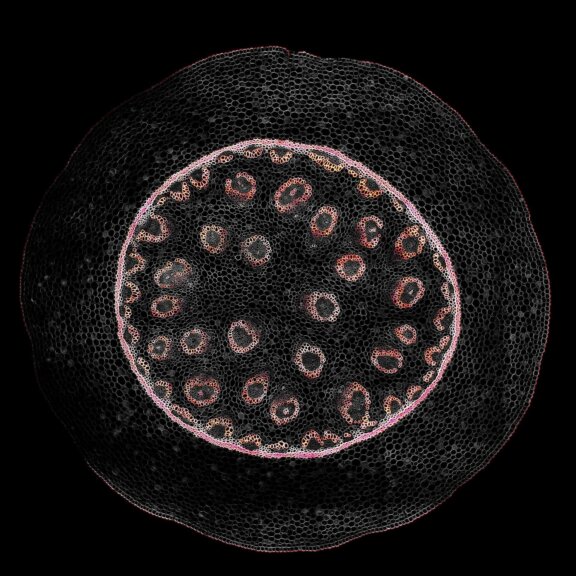
Confocal acquisition of Convallaria rhizome (cross-section) with a 20x oil objective; 3 channels, maximum intensity projection. The image consists of 14 z-planes in 9 by 9 tile pattern comprising a total area of 3.2 mm x 3.2 mm, stitched using SVI Huygens. Shown is a maximum intensity projection.
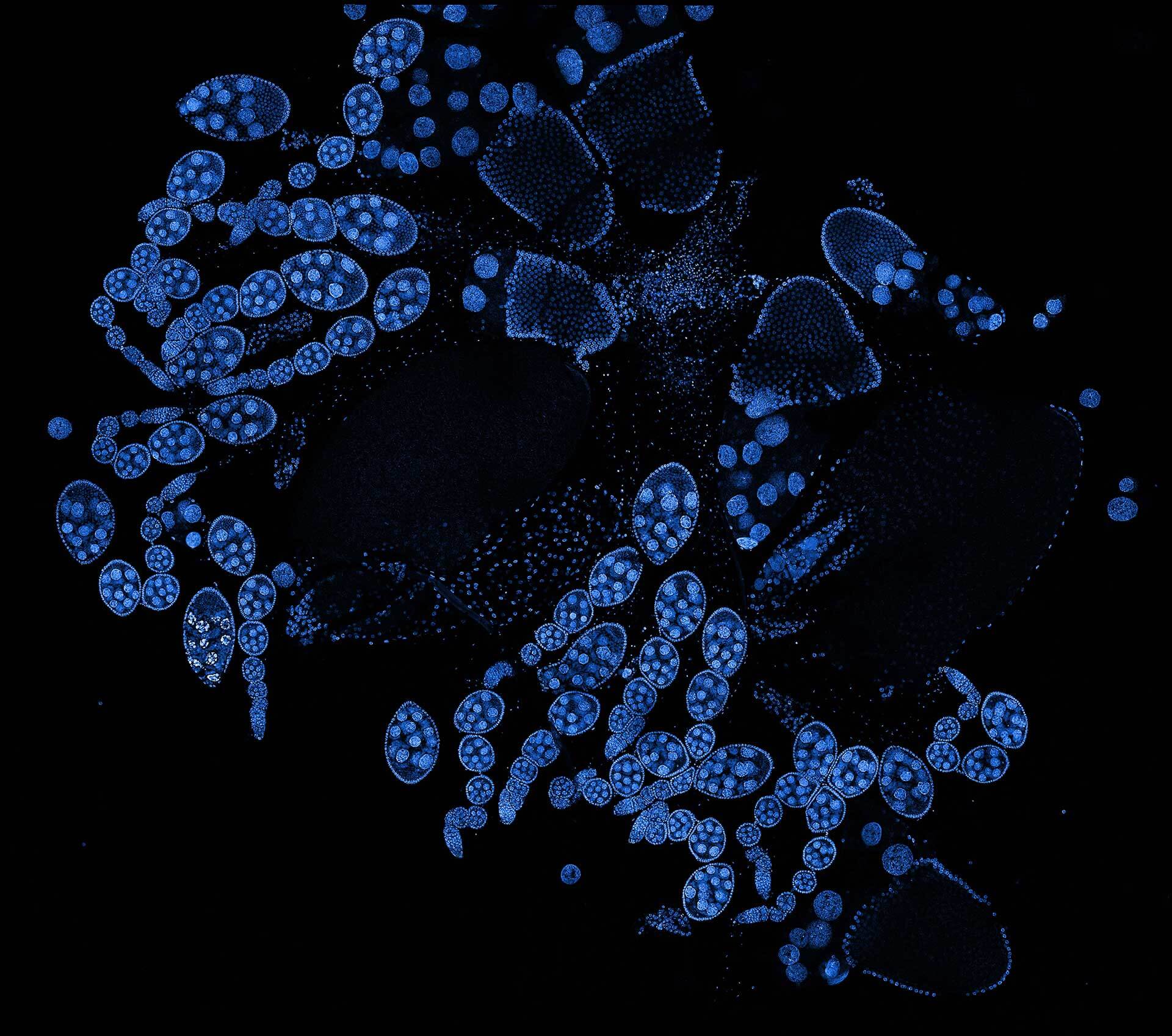
Description
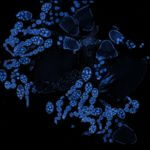
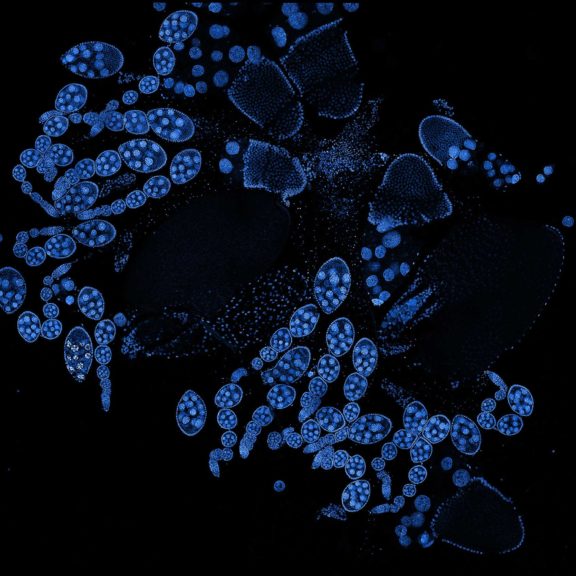
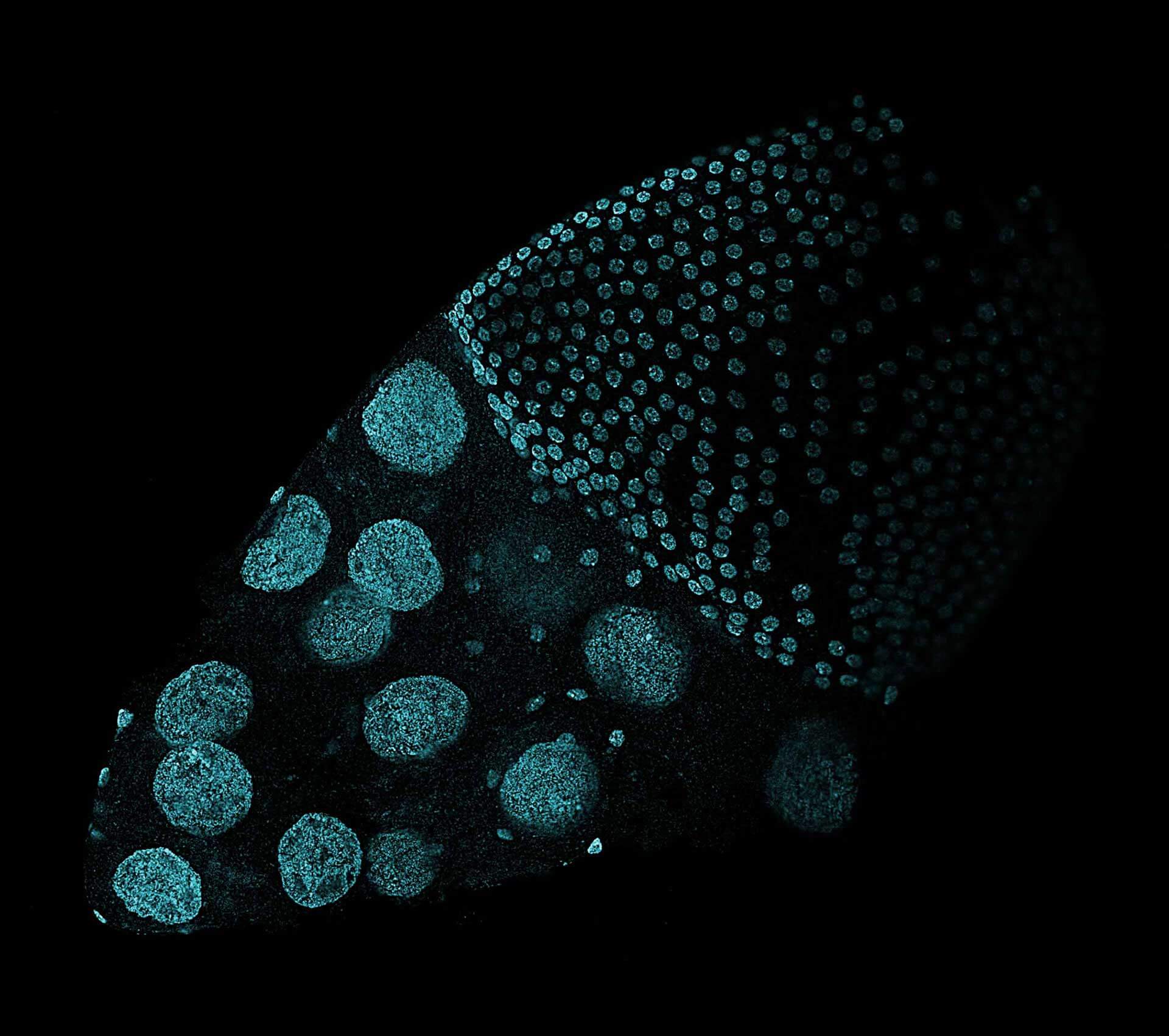
Drosophila ovariole stained with abberior LIVE 560 DNA showing nuclei in different cell types of the egg chamber. Ovaries were dissected from adult female fruit flies and were fixed prior to staining.
Image was acquired with the STEDYCON tiling feature and assembled with the SVI Huygens Stitcher.
Modules:
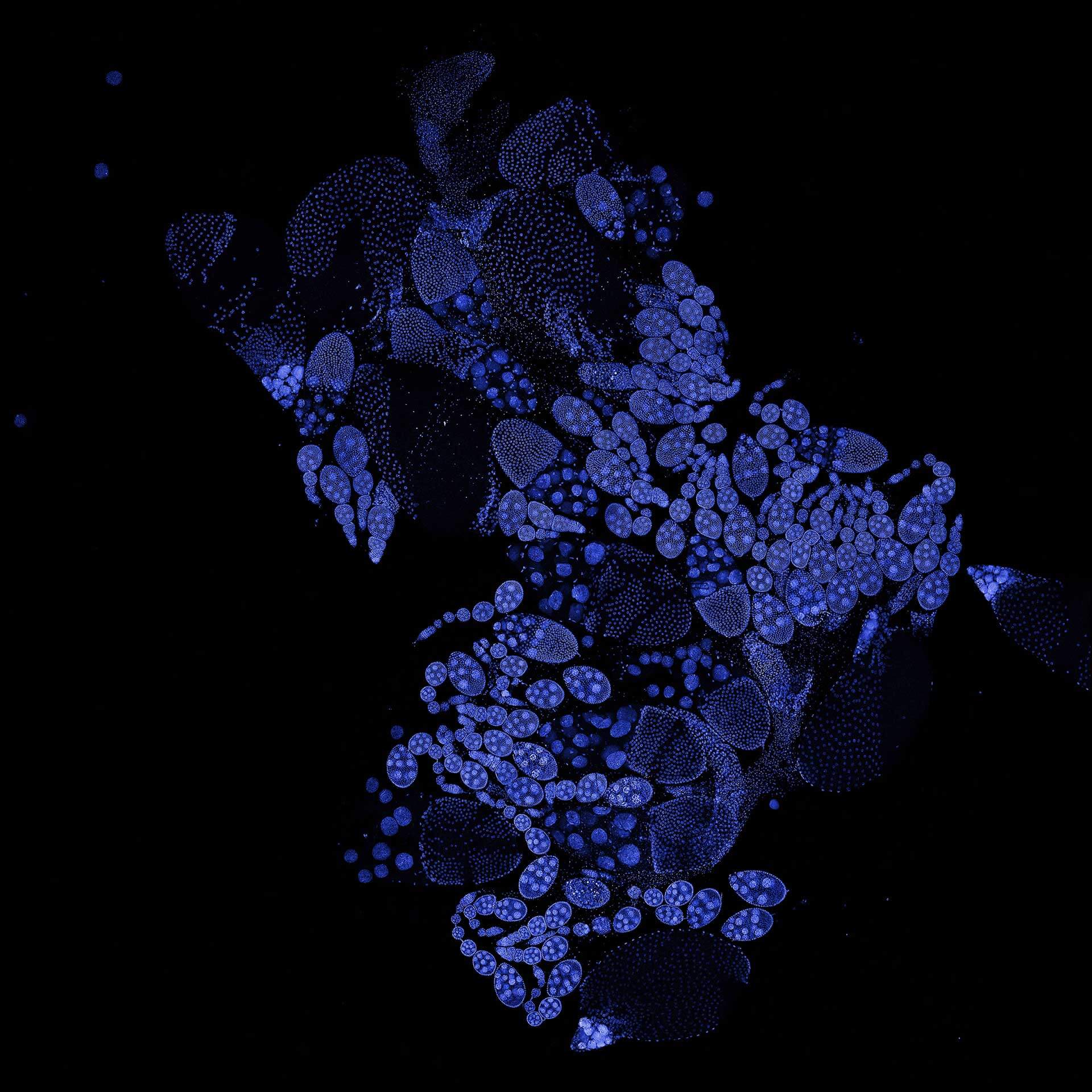
Description
Drosophila ovariole stained with abberior LIVE 560 DNA showing nuclei in different cell types of the egg chamber. Ovaries were dissected from adult female fruit flies and were fixed prior to staining.
Image was acquired with the STEDYCON tiling feature and assembled with the SVI Huygens Stitcher.
Modules:
Description
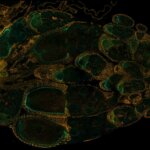
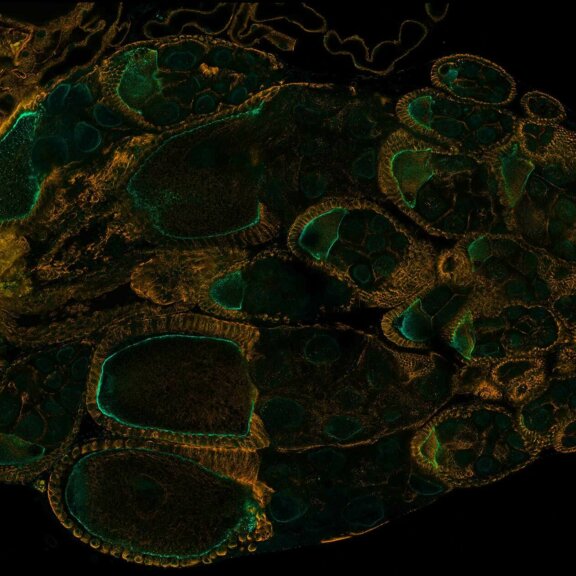
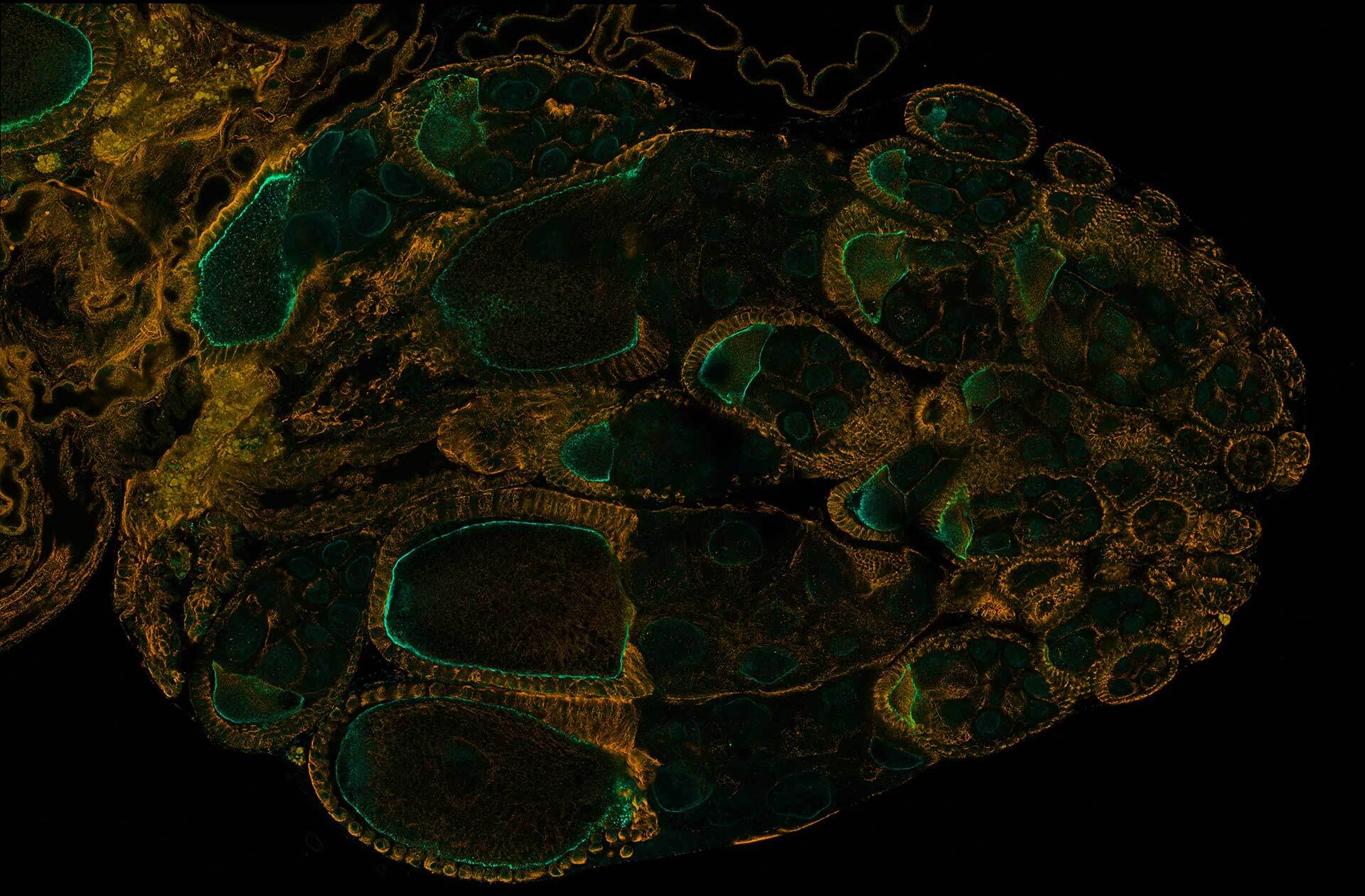
Drosophila ovariole stained with abberior STAR RED phalloidin (actin, red) and nuclear pores in STAR ORANGE (cyan) highlighting different cell types of the egg chamber. Image was acquired with the STEDYCON tiling feature and assembled with the HYGENS stitcher
Description
Drosophila ovariole stained with abberior LIVE 560 DNA, showing the different cell types of the egg chamber. The image was stitched using SVI Huygens.
Description
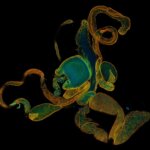
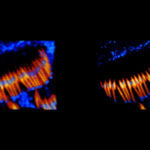
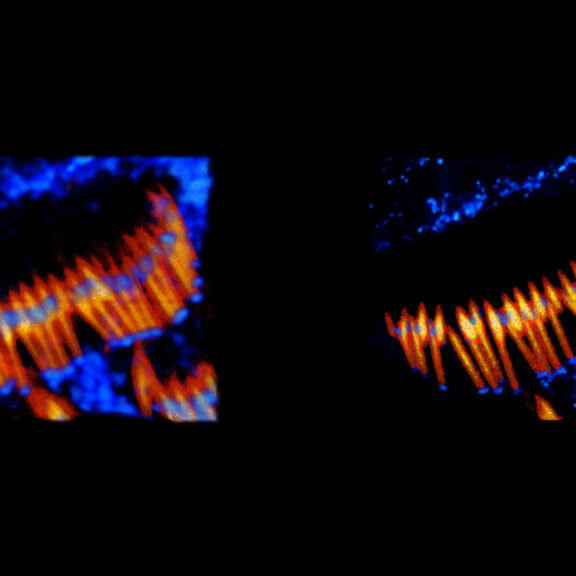
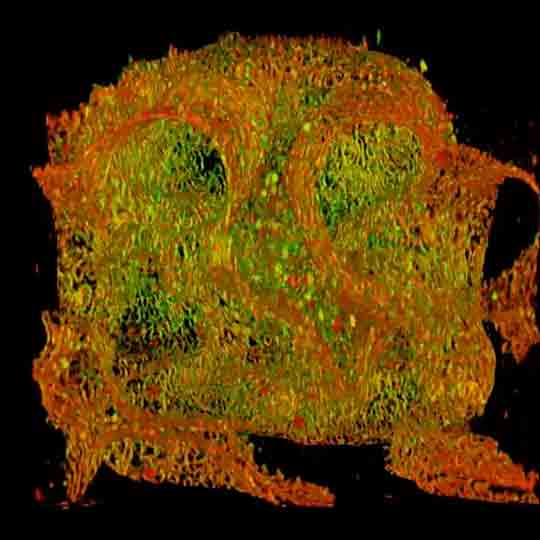
Mouse inner ear hair cells imaged with superresolution in all three dimensions using EASY3D. Animation was generated directly from raw data. Actin stain (abberior STAR 580 phalloidin, blue) and Whirlin immunostain (abberior STAR 635P secondaries, yellow-red). Scalebar 1 µm.
Samples were prepared by Dr. Christian Vogl, InnerEarLab, UMG Göttingen, Germany.
Modules:
Description
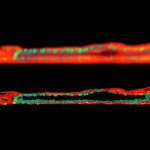
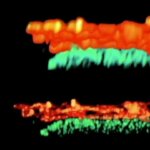
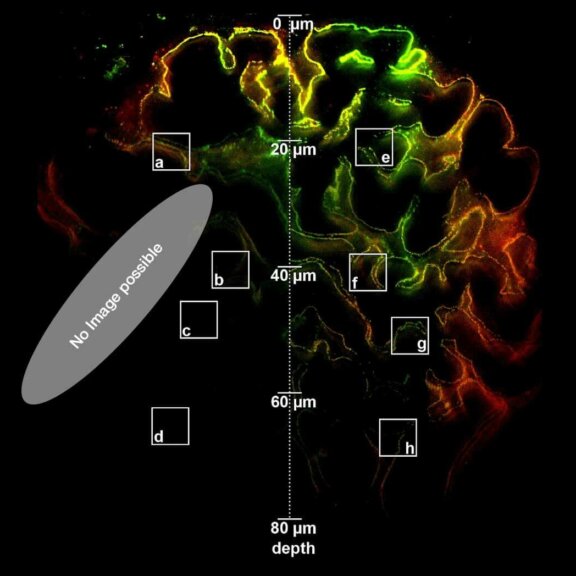
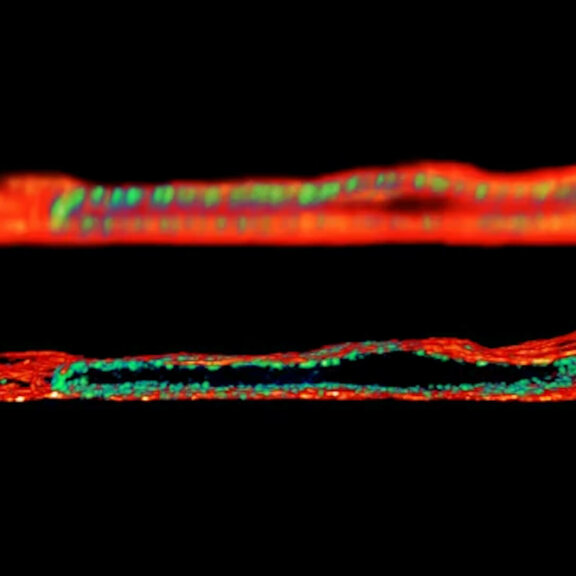
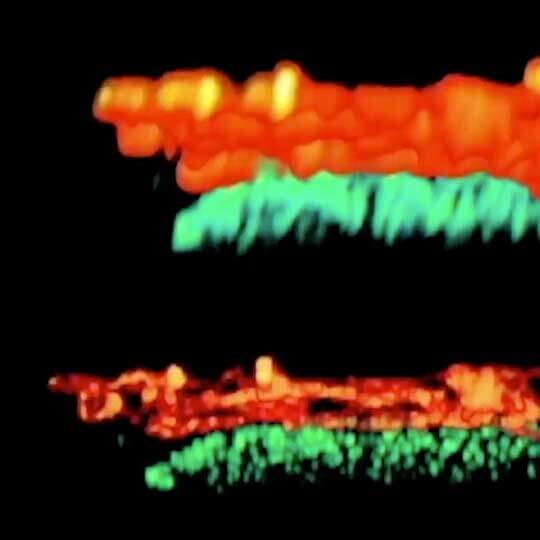
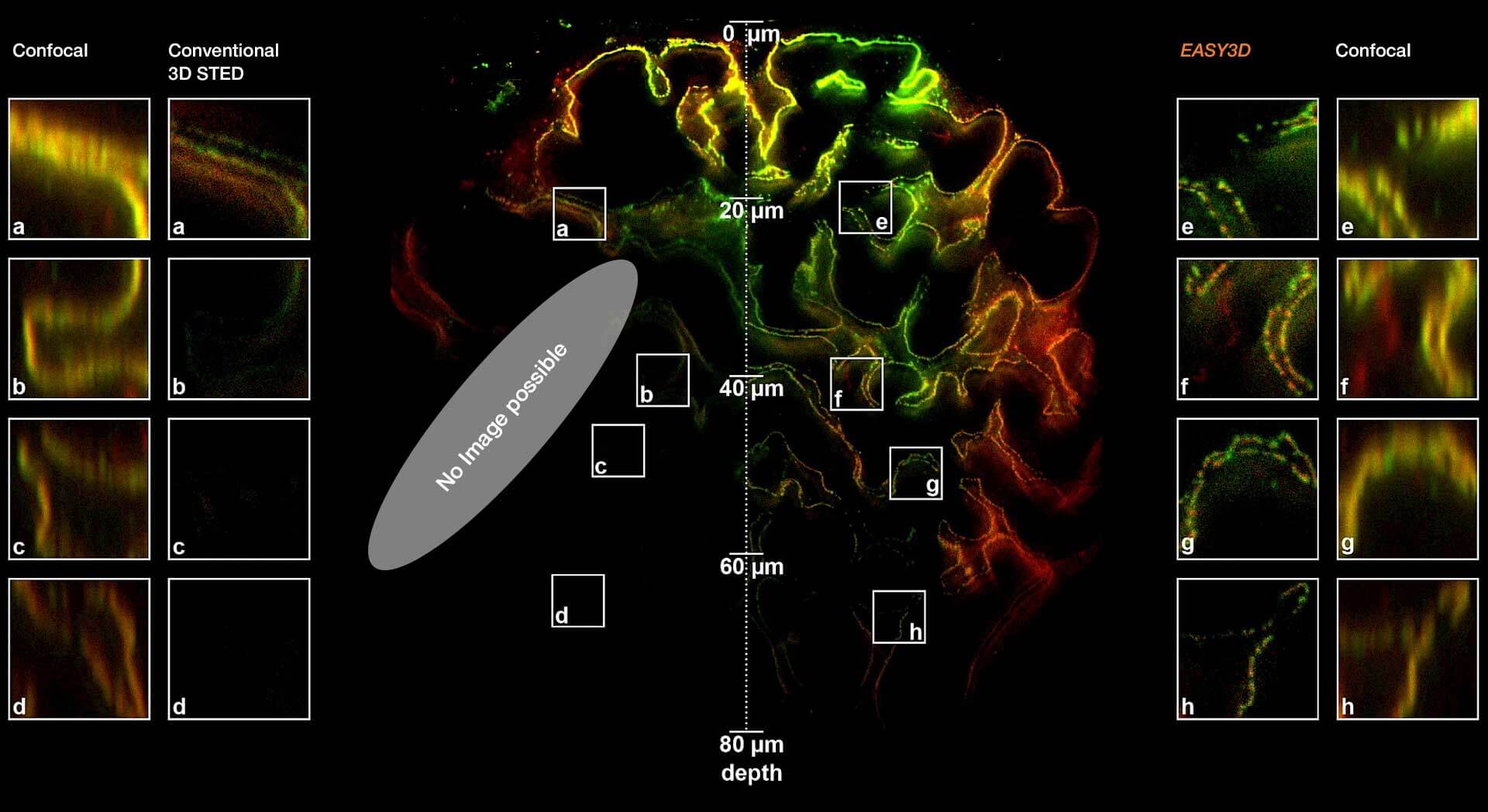
Adaptive optics EASY3D enables deep imaging of cleared adult kidney samples. Comparison of a XZ-slice deep into a renal corpuscle using conventional 3D STED imaging and an EASY3D imaging. EASY3D allowed imaging up to 80 µm into cleared rat kidney tissue using an oil objective. Without adaptive optics, the mismatch between immersion oil and sample leads to complete signal loss. Labels: Nephrin (red, Abberior STAR635P) and Podocin (green, AlexaFluor594).
Sample was prepared by D. Unnersjö Jess and H.G. Blom @ KTH Stockholm, Sweden.
Modules:
Description
Movie through a 2-color EASY3D RESCUE STED volume stack of a cleared adult kidney sample of a rat. Shown is a movie of an EASY3D and RESCUE volume stack of a renal corpuscle showing Nephrin (red, abberior STAR 635P) structures inbetween the Podocin slits (green, AlexaFluor594).
Sample was prepared by D. Unnersjö Jess and H.G. Blom @ KTH Stockholm, Sweden.
Modules:
Description
Surface Rendering of 2-color EASY3D volume of a cleared adult kidney sample of a rat. Shown is a reconstruction of an EASY3D volume stack of a renal corpuscle showing Nephrin (red, abberior STAR 635P) structures inbetween the Podocin slits (green, AlexaFluor594).
Sample was prepared by D. Unnersjö Jess and H.G. Blom @ KTH Stockholm, Sweden.
Modules:



Description
Growth cone at the tip of the axon of a primary hippocampal neuron at 1 day in vitro. Microtubules (Tuj1, abberior STAR 580, red) are bundled in the central-domain suggesting a pausing state. The molecular motor myosin IIB (confocal, Alexa488, blue) is enriched at the transition-zone, along the F-actin arcs. In the peripheral domain actin forms bundles in the filopodia (Phalloidin, abberior STAR 635, green). Sample courtesy: Elisa D'Este, Max Planck Institute for Biophysical Chemistry, Göttingen, Germany.
Description
3-color STED image of primary hippocampal neurons. Please note the characteristic ~190 nm beta II spectrin periodicity along distal axons (green) which is only visible in the STED image. Labelled structures: beta II spectrin (green, abberior STAR 635P), Bassoon (red, abberior STAR 580), Actin cytoskeleton (blue, phalloidin, Oregon Green 488). Imaged with abberior Expert Line with 595nm and 775nm STED laser. Sample was prepared by Elisa D’Este @ MPIBPC, Göttingen.
Modules:
Description
3-color STED imaging: active zones at the Drosophila larval neuromuscular junction immunostained for Bruchpilot and two other proteins.
Two superresolution channels (magenta, yellow) using a 775nm STED laser & one superresolution channel using a 595nm STED laser.
Samples by M. Lenz & M. Landgraf (University of Cambridge, UK).
Modules:
Modules:
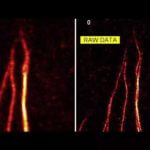
Description
Spherical aberrations arising from index mismatch between sample and immersion are corrected for, as well as higher-order, sample-induced aberrations.
Without Adaptive Optics, excitation laser power is typically increased with focus depth in order to compensate loss of signal due to aberrations. Adaptive Optics preserves resolution and brightness deep inside thick samples and enables imaging at low light levels.
Sample courtesy of Amelie Cabirol and Albrecht Haase, University of Trento.
Modules:
Description
Confocal acquisition of Convallaria rhizome (cross-section) with a 20x oil objective; 3 channels, maximum intensity projection. The image consists of 14 z-planes in 9 by 9 tile pattern comprising a total area of 3.2 mm x 3.2 mm, stitched using SVI Huygens. Shown is a maximum intensity projection.
Description
Drosophila ovariole stained with abberior LIVE 560 DNA showing nuclei in different cell types of the egg chamber. Ovaries were dissected from adult female fruit flies and were fixed prior to staining.
Image was acquired with the STEDYCON tiling feature and assembled with the SVI Huygens Stitcher.
Modules:
Description
Drosophila ovariole stained with abberior LIVE 560 DNA showing nuclei in different cell types of the egg chamber. Ovaries were dissected from adult female fruit flies and were fixed prior to staining.
Image was acquired with the STEDYCON tiling feature and assembled with the SVI Huygens Stitcher.
Modules:
Description
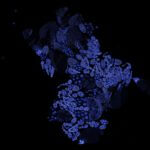
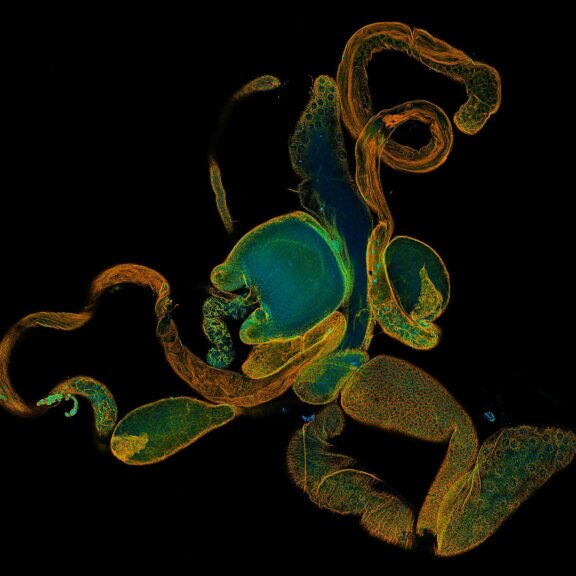
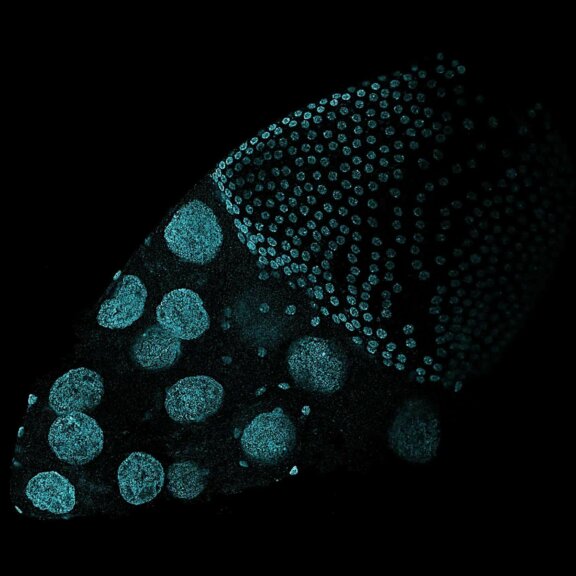
Tiled and stitched image of drosophila testis. Sample by J. Rehman.
Description
Drosophila ovariole stained with abberior STAR RED phalloidin (actin, red) and nuclear pores in STAR ORANGE (cyan) highlighting different cell types of the egg chamber. Image was acquired with the STEDYCON tiling feature and assembled with the HYGENS stitcher
Description
Drosophila ovariole stained with abberior LIVE 560 DNA, showing the different cell types of the egg chamber. The image was stitched using SVI Huygens.
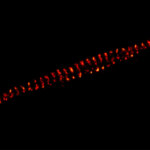
Description
3D superresolution volume image of active zones at the Drosophila larval neuromuscular junction. Immunostaining for Bruchpilot and a second protein.
Recorded using EASY3D at 775nm. Samples by M. Lenz & M. Landgraf (University of Cambridge, UK).
Modules:
Description
Mouse inner ear hair cells imaged with superresolution in all three dimensions using EASY3D. Animation was generated directly from raw data. Actin stain (abberior STAR 580 phalloidin, blue) and Whirlin immunostain (abberior STAR 635P secondaries, yellow-red). Scalebar 1 µm.
Samples were prepared by Dr. Christian Vogl, InnerEarLab, UMG Göttingen, Germany.
Modules:
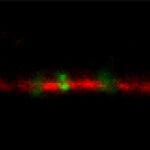
Description
Adaptive optics EASY3D enables deep imaging of cleared adult kidney samples. Comparison of a XZ-slice deep into a renal corpuscle using conventional 3D STED imaging and an EASY3D imaging. EASY3D allowed imaging up to 80 µm into cleared rat kidney tissue using an oil objective. Without adaptive optics, the mismatch between immersion oil and sample leads to complete signal loss. Labels: Nephrin (red, Abberior STAR635P) and Podocin (green, AlexaFluor594).
Sample was prepared by D. Unnersjö Jess and H.G. Blom @ KTH Stockholm, Sweden.
Modules:
Description
Nuclear pore complexes (NUP) and tubulin imaged with confocal and EASY3D-STED microscopy.
Modules:
Description
Movie through a 2-color EASY3D RESCUE STED volume stack of a cleared adult kidney sample of a rat. Shown is a movie of an EASY3D and RESCUE volume stack of a renal corpuscle showing Nephrin (red, abberior STAR 635P) structures inbetween the Podocin slits (green, AlexaFluor594).
Sample was prepared by D. Unnersjö Jess and H.G. Blom @ KTH Stockholm, Sweden.
Modules:
Description
Surface Rendering of 2-color EASY3D volume of a cleared adult kidney sample of a rat. Shown is a reconstruction of an EASY3D volume stack of a renal corpuscle showing Nephrin (red, abberior STAR 635P) structures inbetween the Podocin slits (green, AlexaFluor594).
Sample was prepared by D. Unnersjö Jess and H.G. Blom @ KTH Stockholm, Sweden.
Modules:
Description
Two-color EASY3D-STED image of tubulin and giantin versus its confocal low-resolution counterpart.