Sample gallery
Fluorescence imaging, whether at confocal, STED or MINFLUX resolution, guarantees unique insights into the function and structure of life at the molecular level. Besides the scientific information content, some sample portraits provide simply beautiful images. Enjoy browsing our sample gallery.
the fine art of science
Description
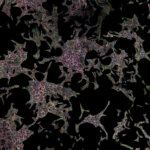
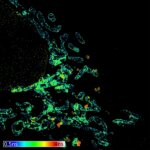
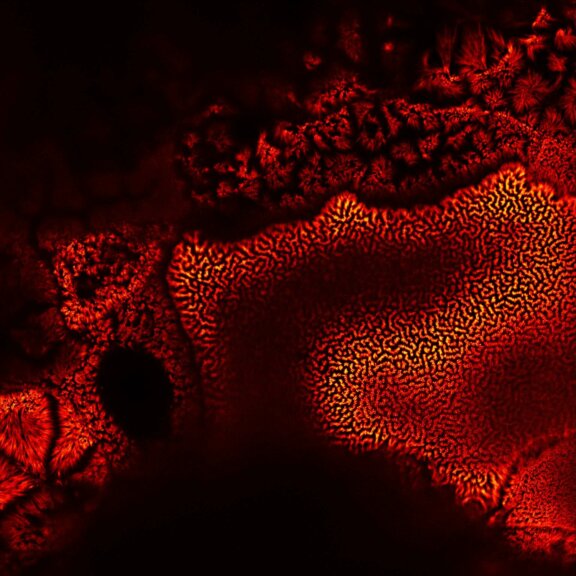
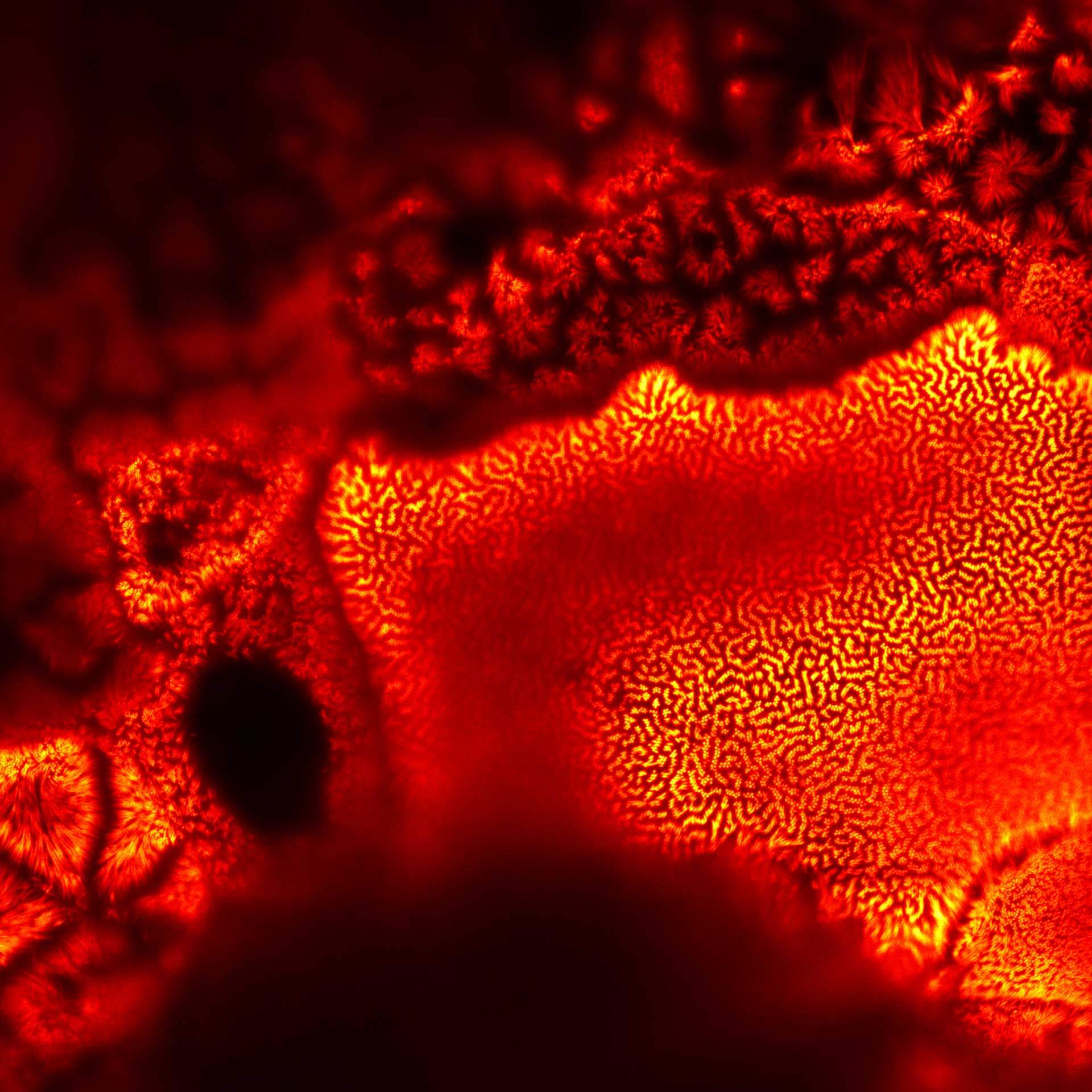
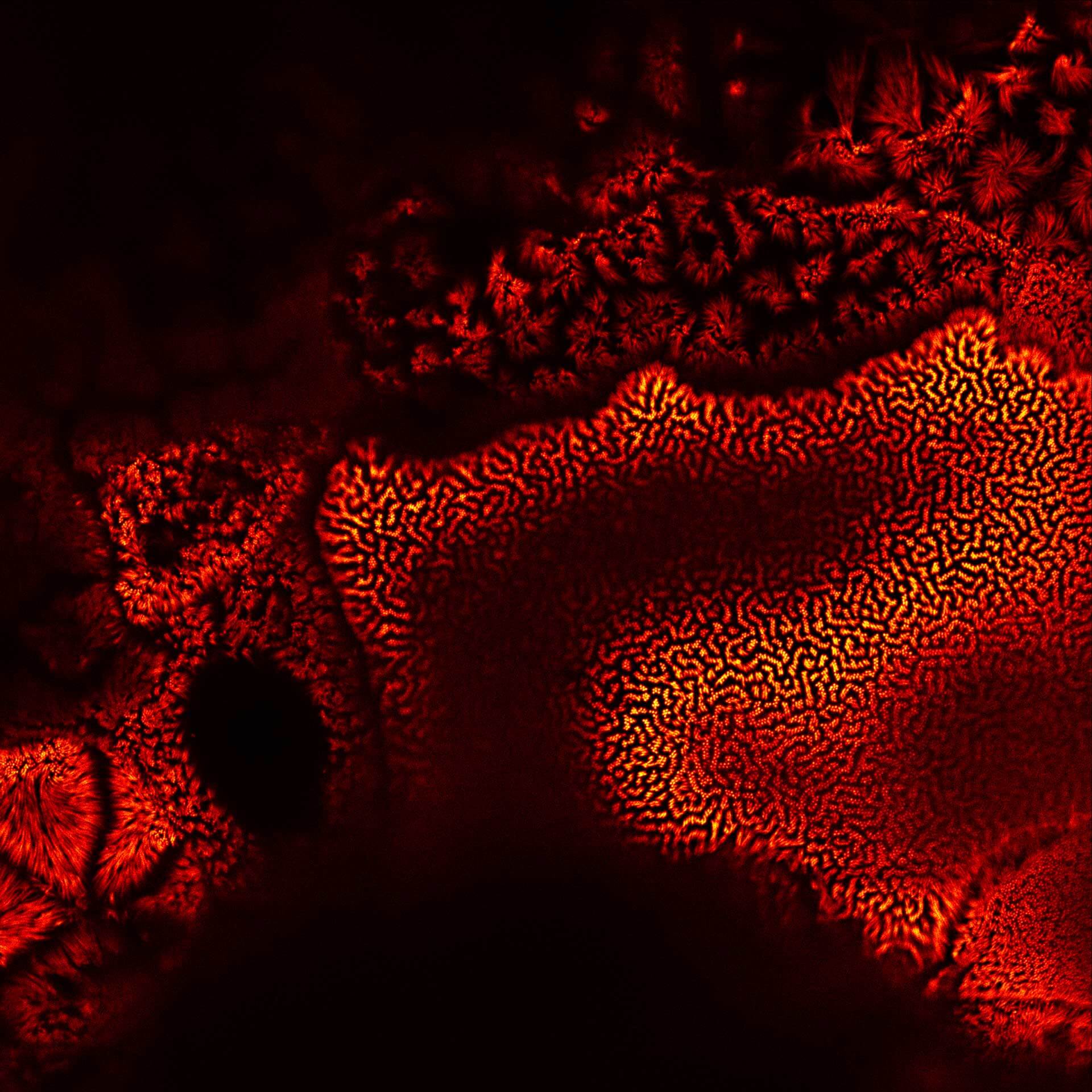
Actin in microvilli of Caco2 cells. Fixed cells were stained with abberior STAR RED phalloidin. MATRIX detection removes background signal and dramatically improves optical sectioning for 2D-STED.
Samples by Prof. Dorothee Günzel & Roman Mannweiler, Institut für Klinische Physiologie, Charité - Universitätsmedizin Berlin.
Modules:
Description
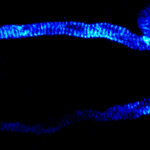
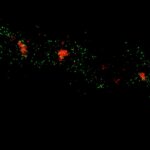
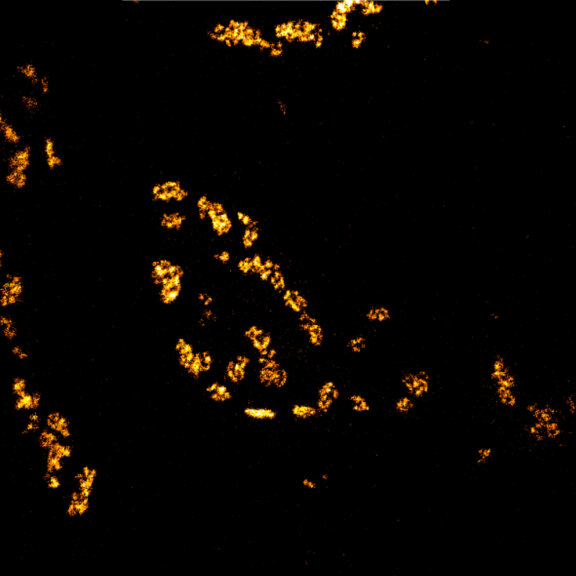
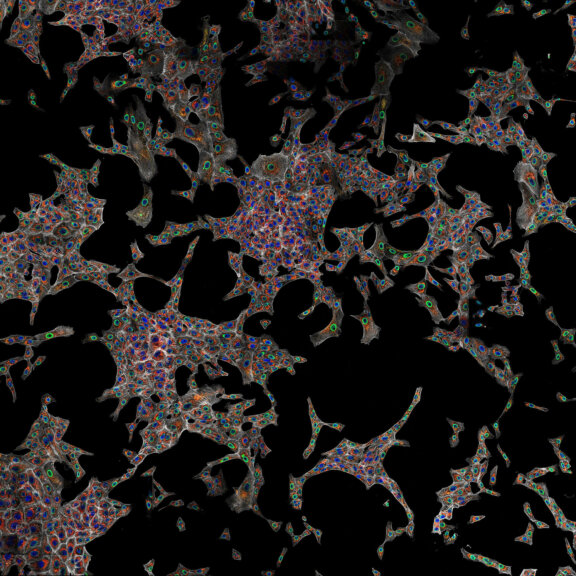
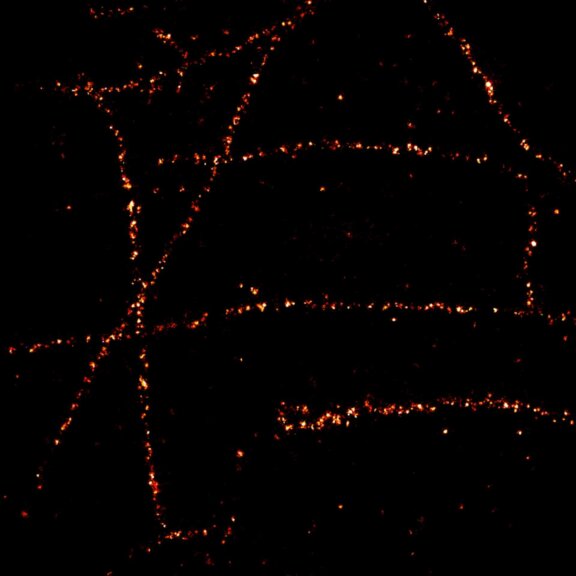
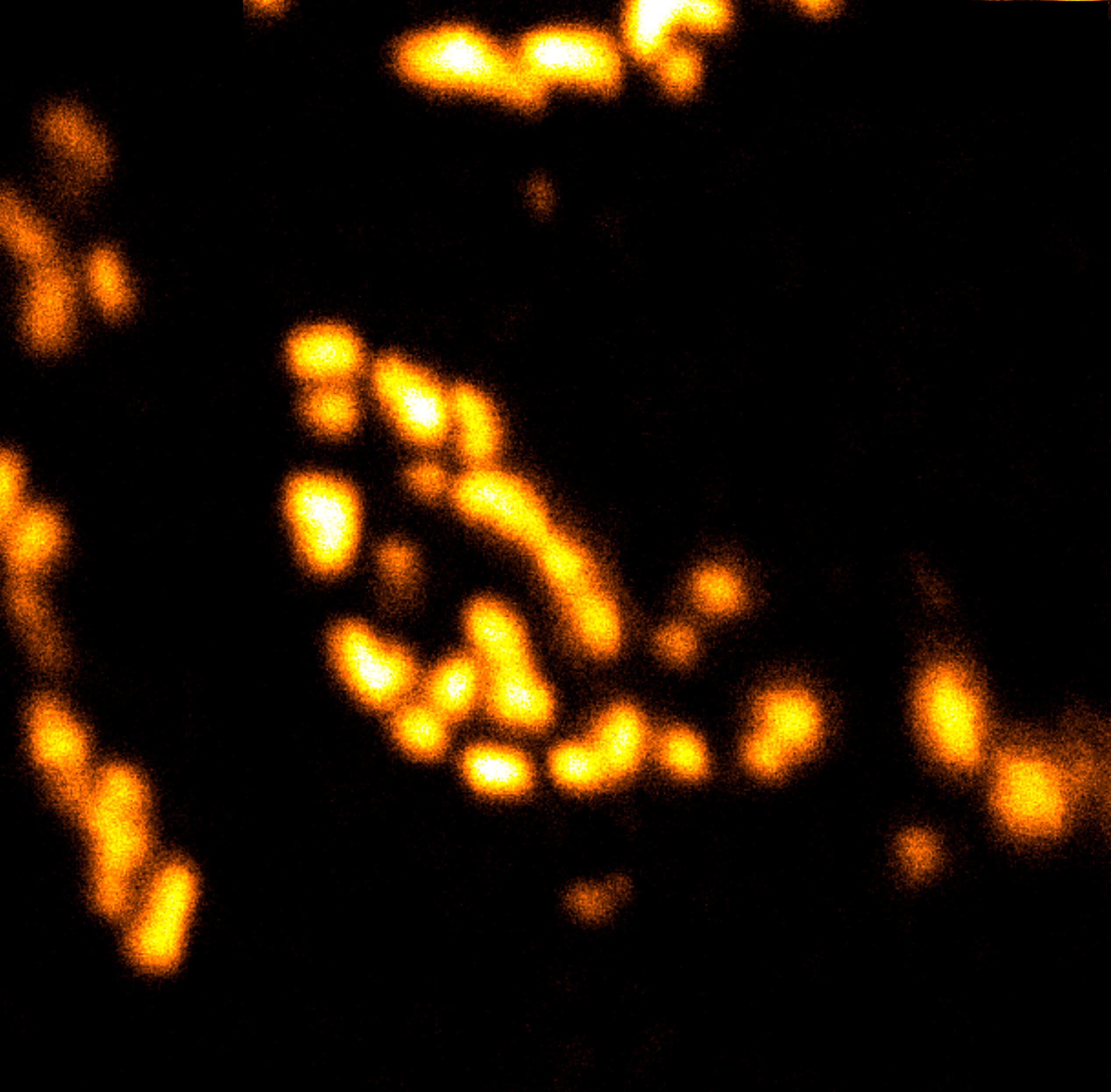
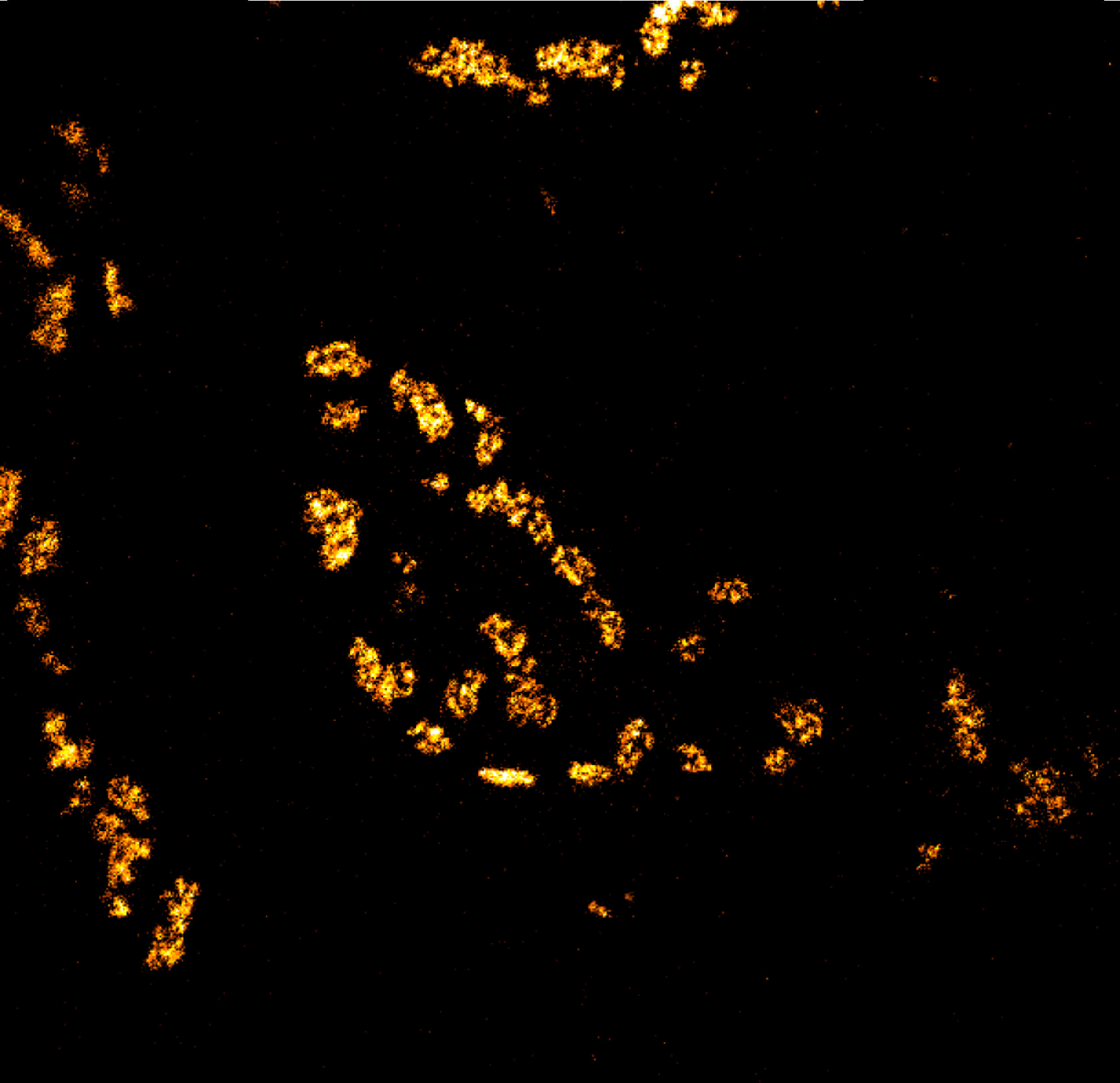
Body wall preparation of a 3rd instar Drosophila melanogaster larva. Synapses at the neuromuscular junction are visualized by immunostaining against Bruchpilot (Brp), an integral component of active zones in Drosophila. Images were acquired on a STEDYCON attached to an Olympus IX81. Images were kindly provided by Dr. Nadine Ehmann, Institute of Physiology, Neurophysiology, University of Würzburg.
Description
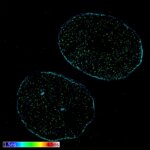
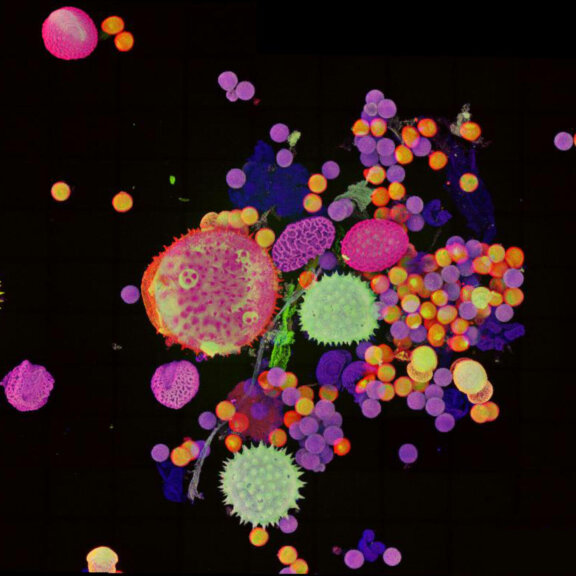
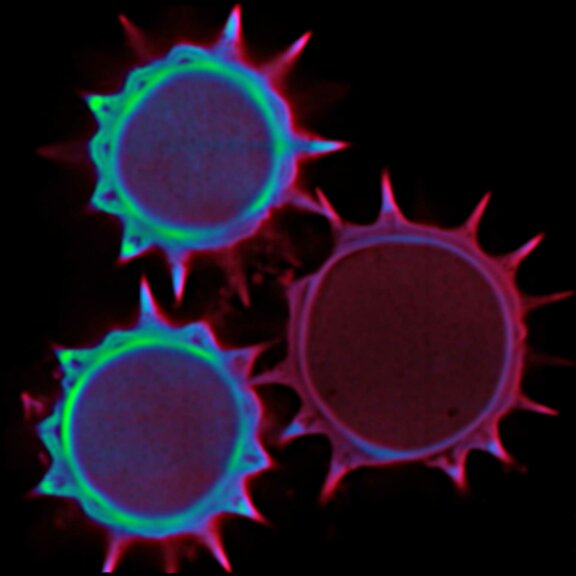
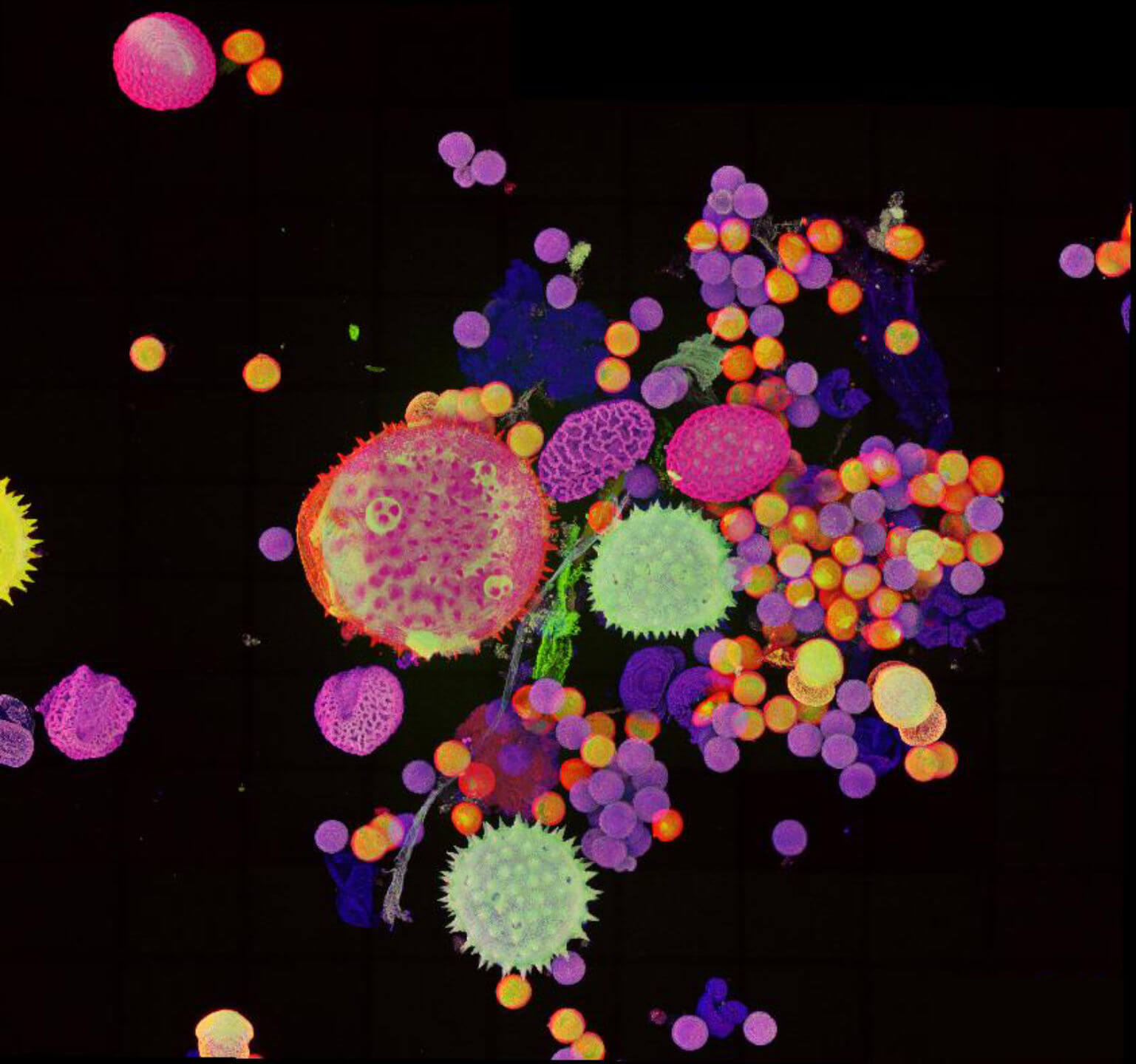
Pollen autofluorescence imaged with a STEDYCON on an IX83 microscope frame. z-stacks on 95 tiles were acquired and stitched with SVI Huygens.
Description
Immunolabelling of βIV-spectrin, a scaffolding protein of the axon initial segment, using a self-made primary antibody and Alexa 594 secondary antibody. Cryo-section of mouse neocortex cut at 20 µm. Images were acquired using a STEDYCON attached to a Zeiss AxioImager Z2. The sample was kindly provided by Dr. Maren Engelhardt, Institute of Neuroanatomy, Medical Faculty Mannheim, Heidelberg University.
Description
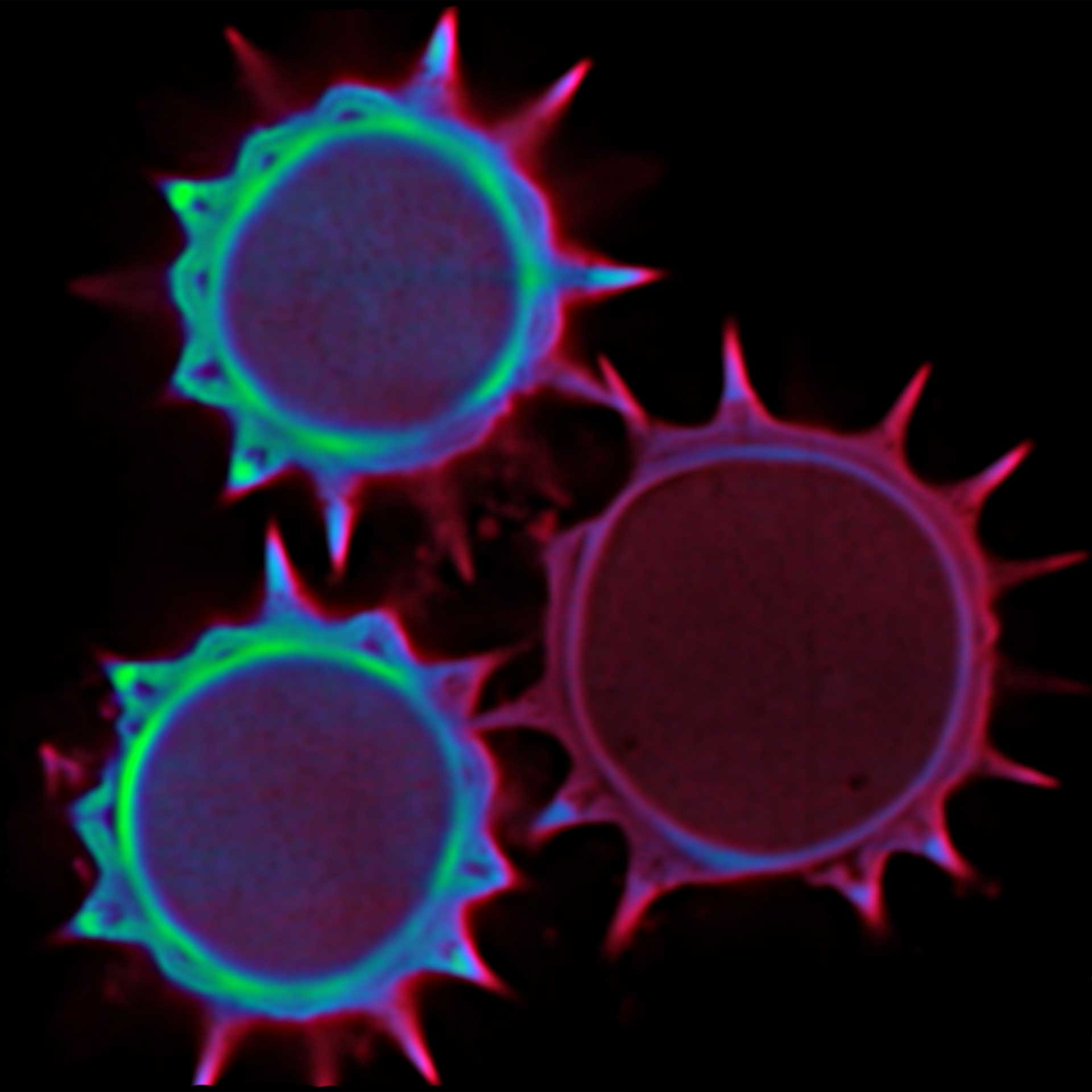
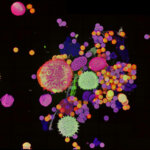
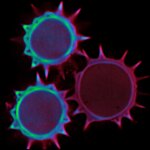
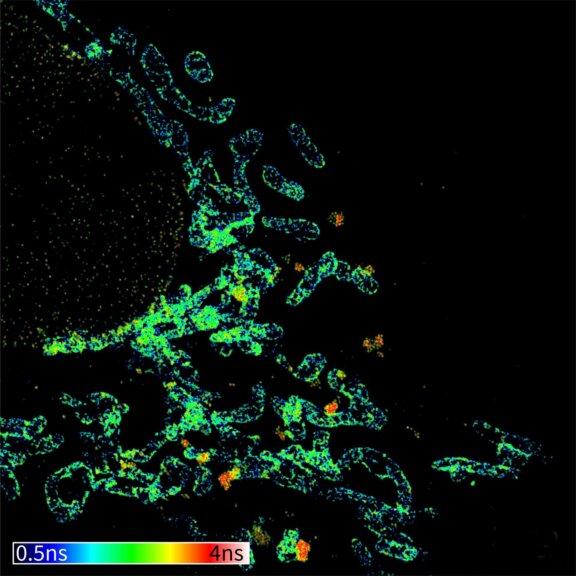
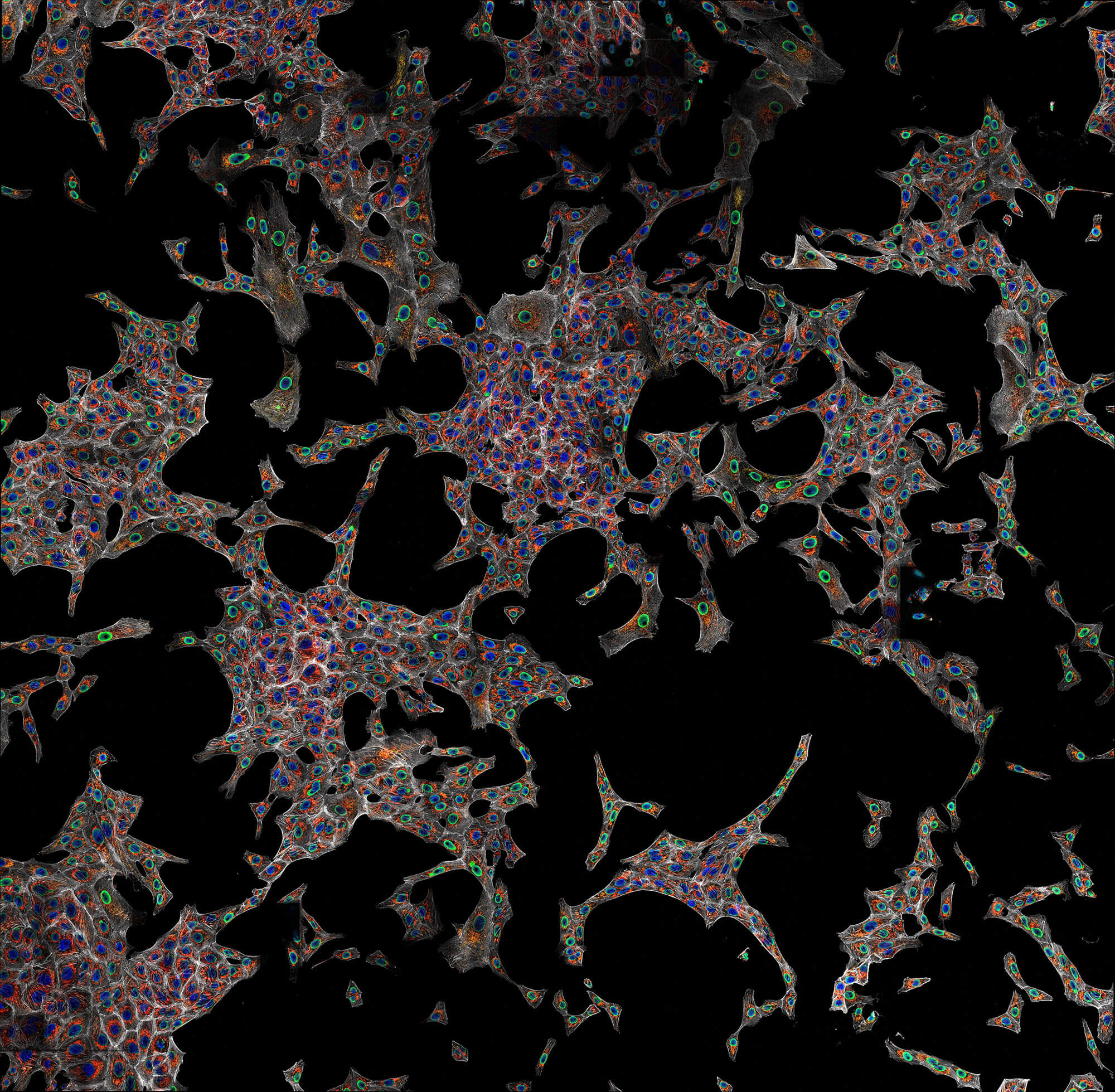
Confocal imaging of a large sample region. Shown is a 2.8 mm x 2.5 mm region, acquired using 35 by 29 tiles using the STEADYFOCUS and stitched with SVI Huygens. Sample: mammalian cells immunolabelled for the mitochondrial protein TOM20 (abberior STAR RED, red), double-stranded DNA to visualize mitochondrial DNA (abberior STAR ORANGE, green), phalloidin to label F-actin (abberior STAR GREEN, gray), and DAPI to label nuclei. Sample preparation by abberior GmbH.
Description
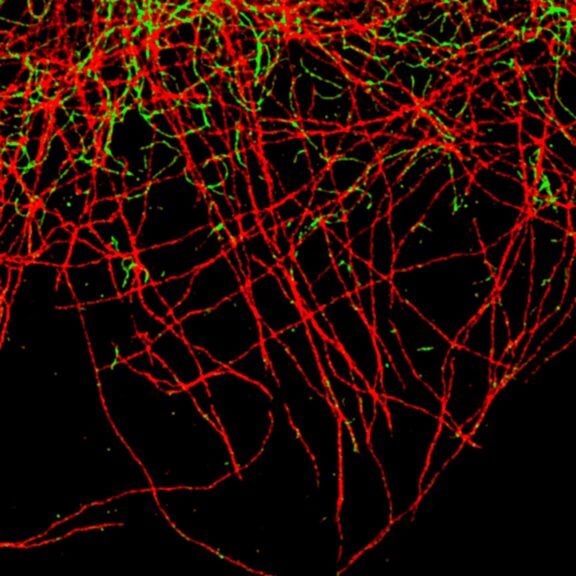
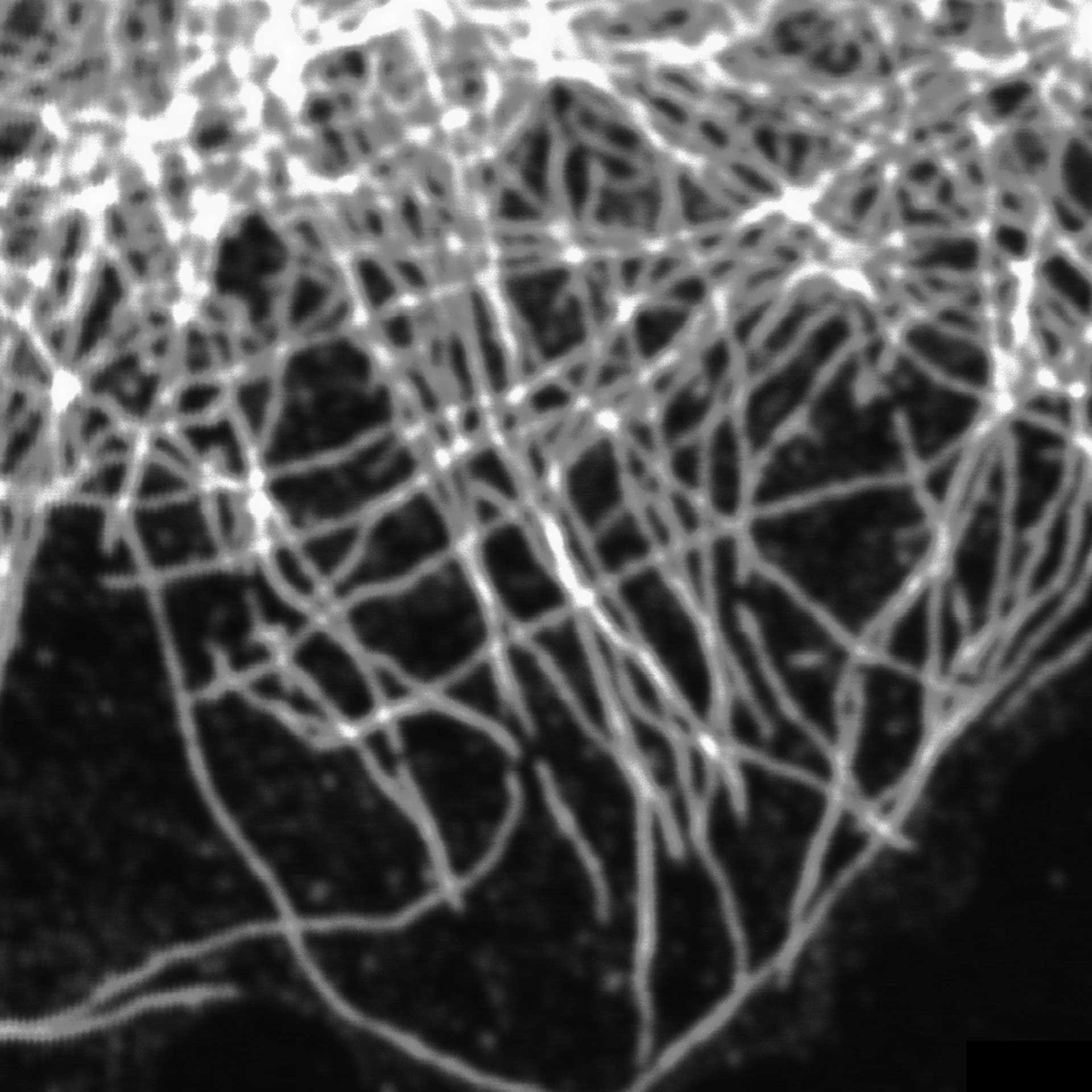
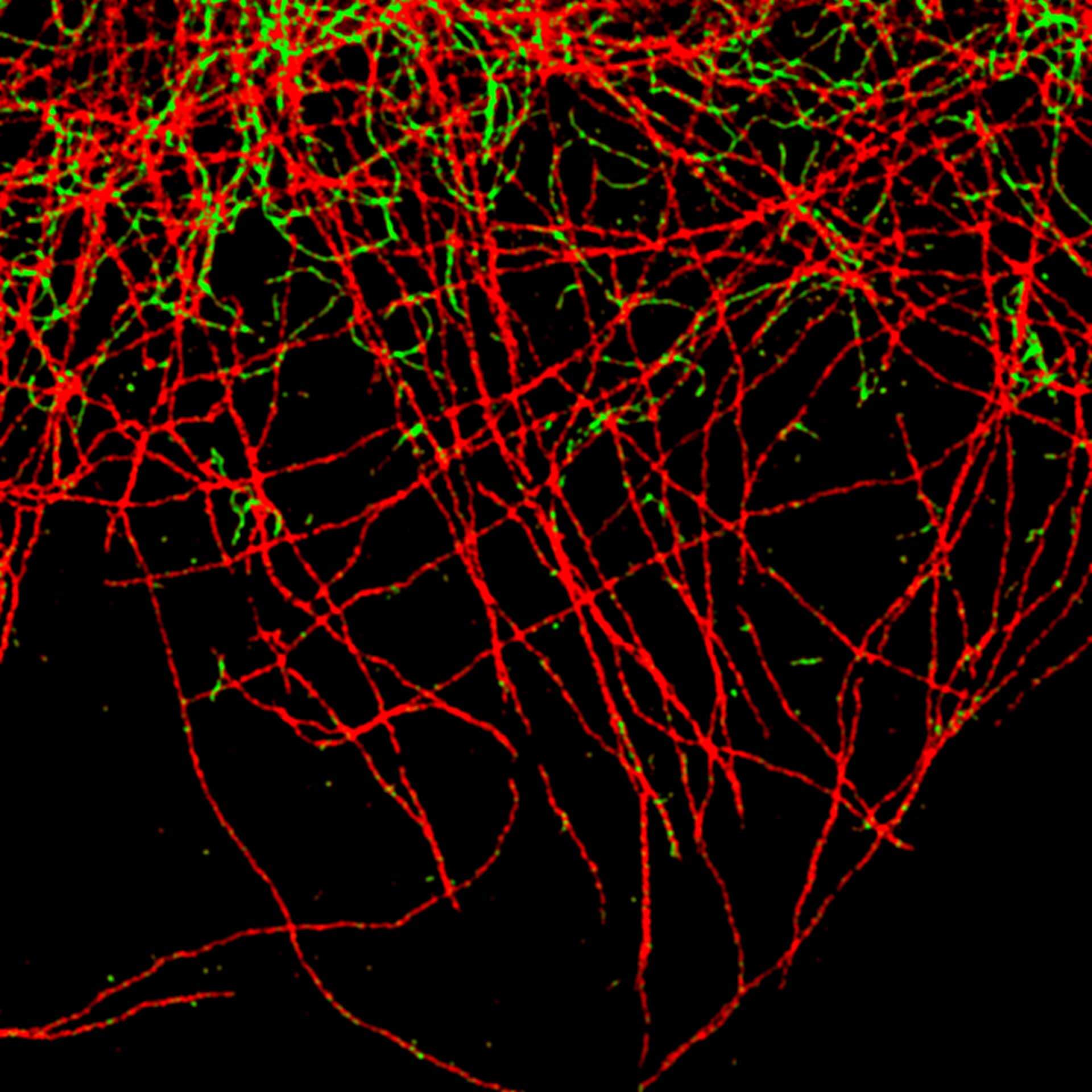
TIMEBOW image of tubulin labeled with Atto647N and vimentin labeled with abberior STAR 635P. The two labels (Atto647N and abberior STAR 635P) have the same excitation spectra, but different lifetimes. Tubulin (red, short lifetime) and vimentin (green, long lifetime) can successfully be separated in the TIMEBOW STED recording via their lifetimes.
Modules:
Description
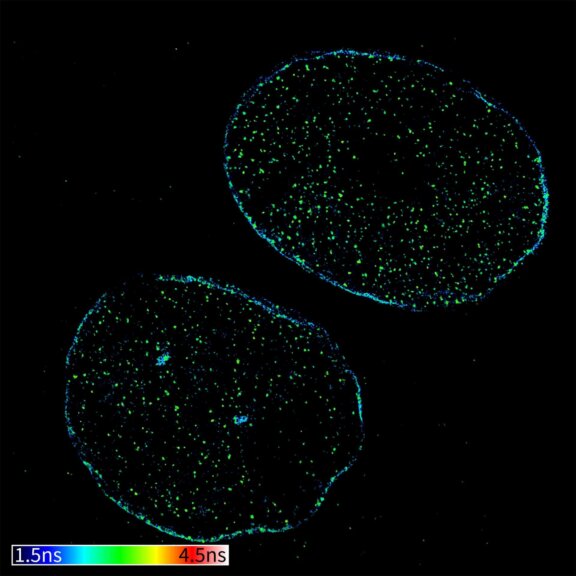
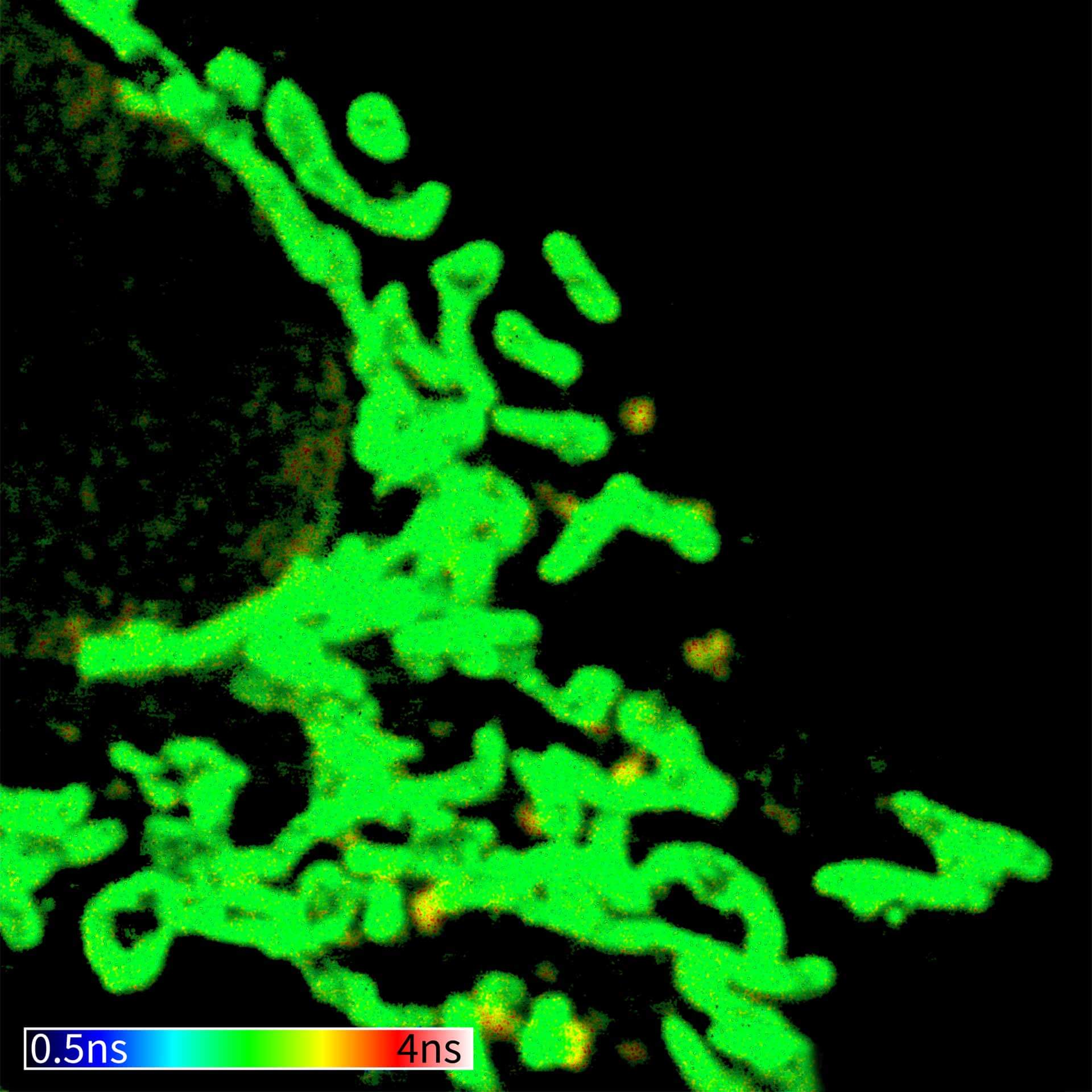
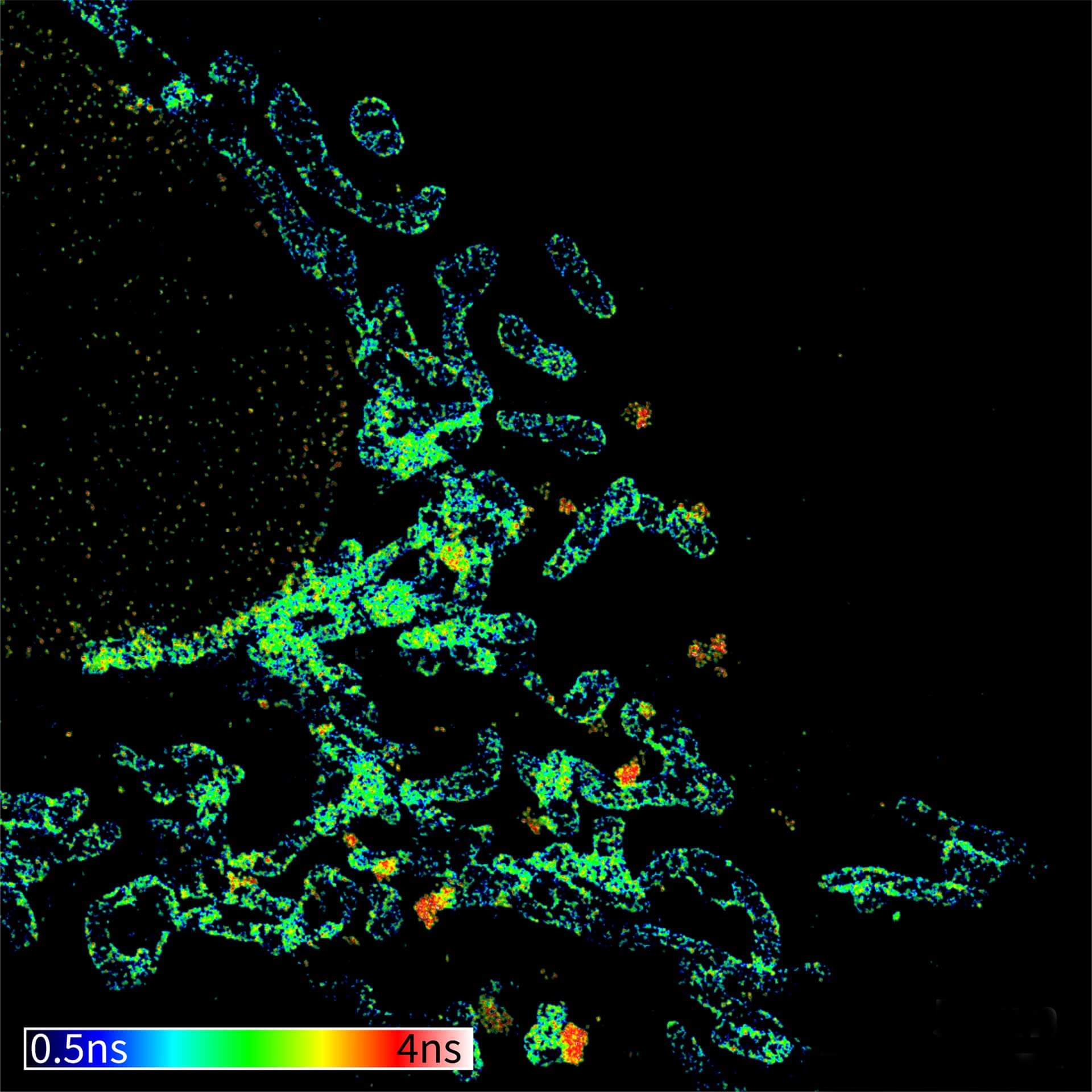
TIMEBOW image of mammalian cells labeled with antibodies against Tom20, ATTO 647N and NUP153, abberior STAR 635P. Note that nuclear pore (NUP) complex subunits appear to be localized in the cytoplasm, which is due to the NUP import pathway into the nuclear membrane.
Modules:
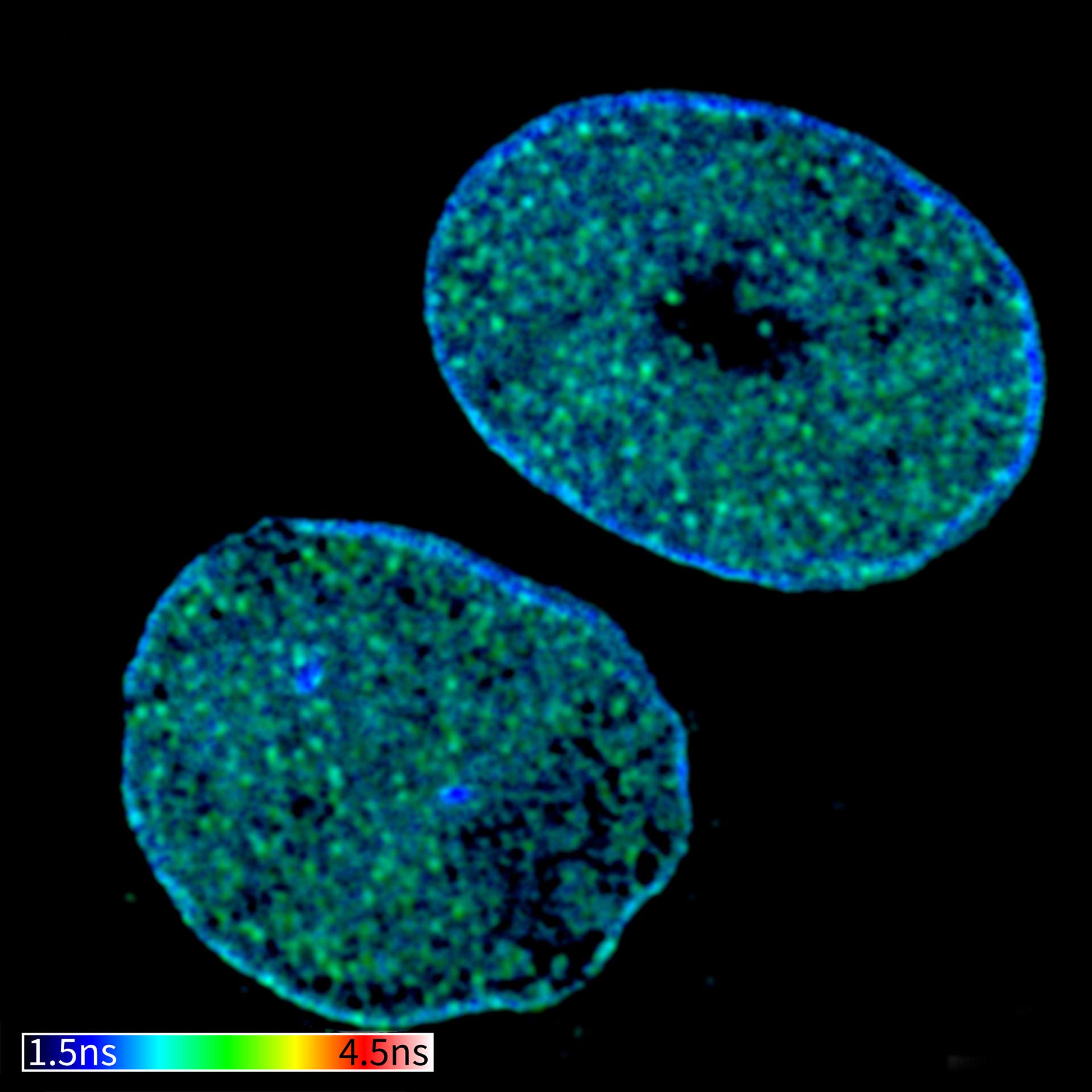
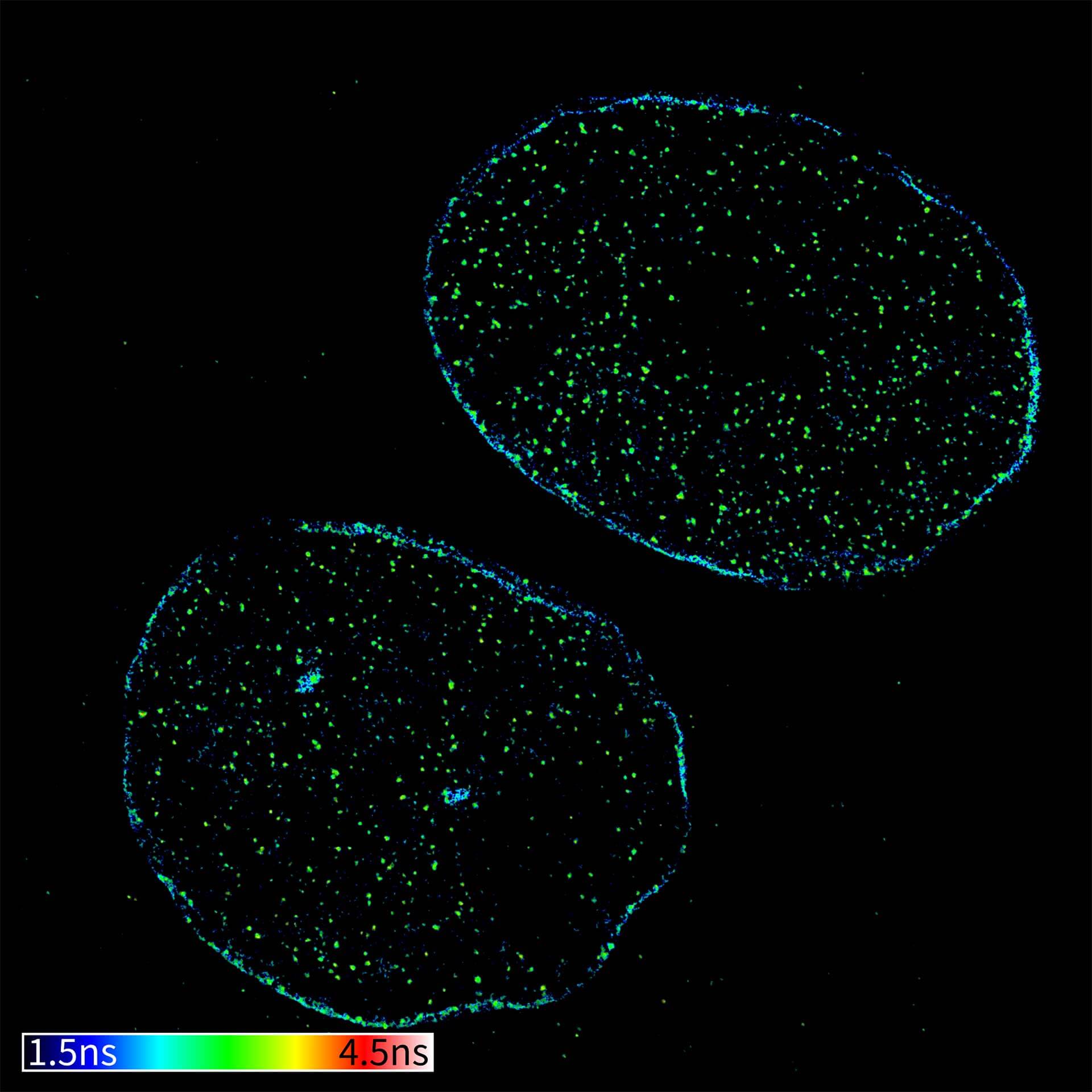
Description
TIMEBOW confocal and TIMEBOW STED image of mammalian cells labeled with antibodies against lamin, ATTO 647N and dsDNA, abberior STAR 635P.
Modules:
Description
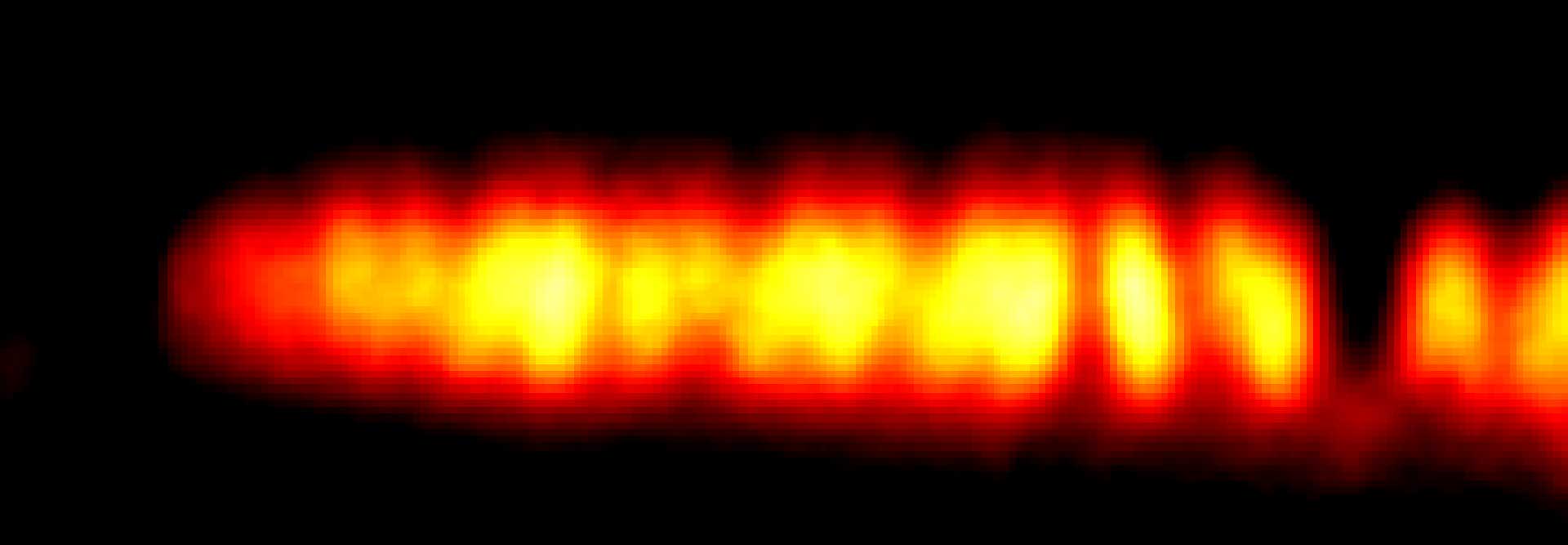
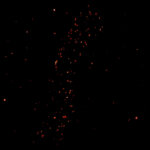
2D tracking of a lipid-coupled Atto 647N molecule embedded in a supported lipid bilayer. The movement of the labeled lipid was tracked with a frequency of up to 10 kHz.

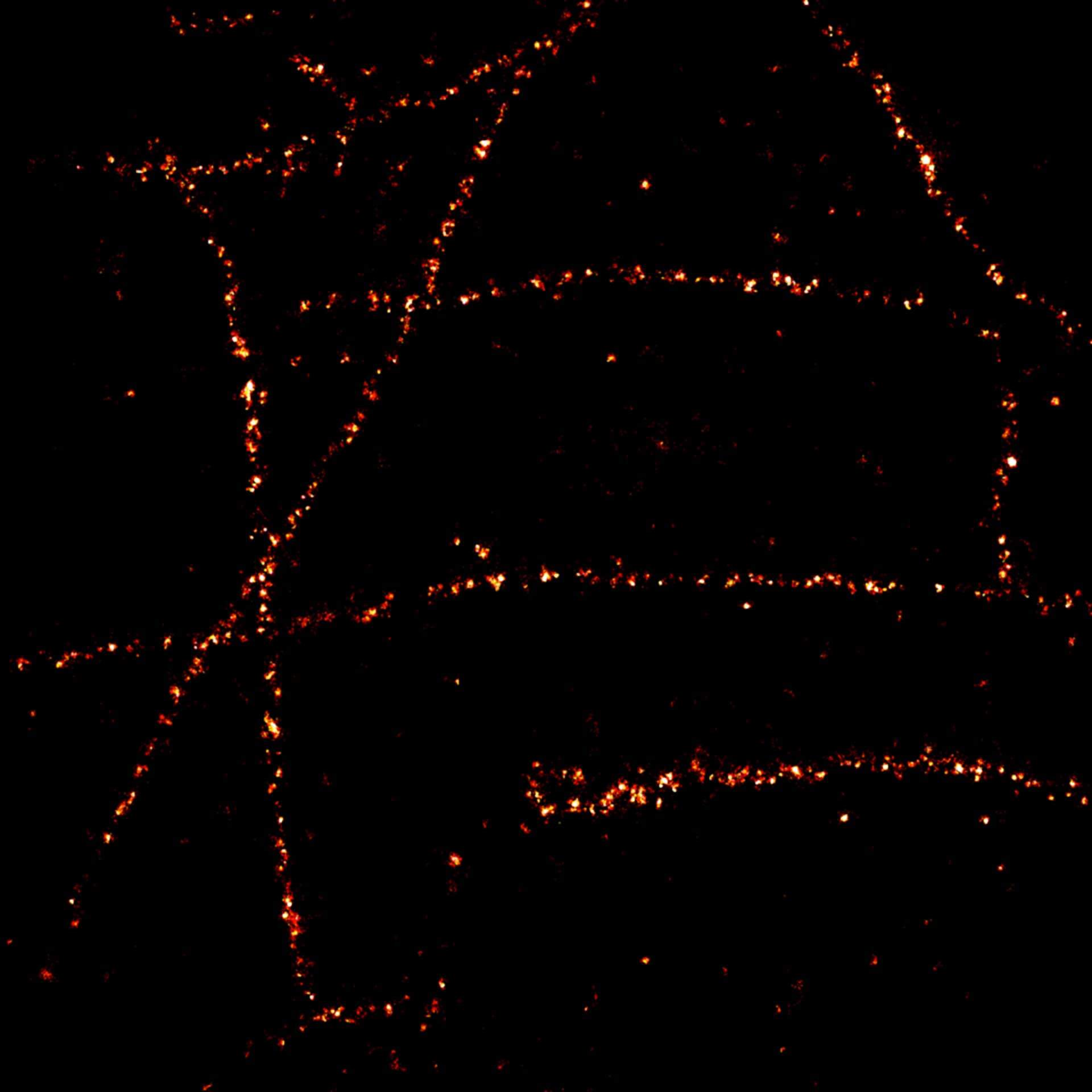
Description
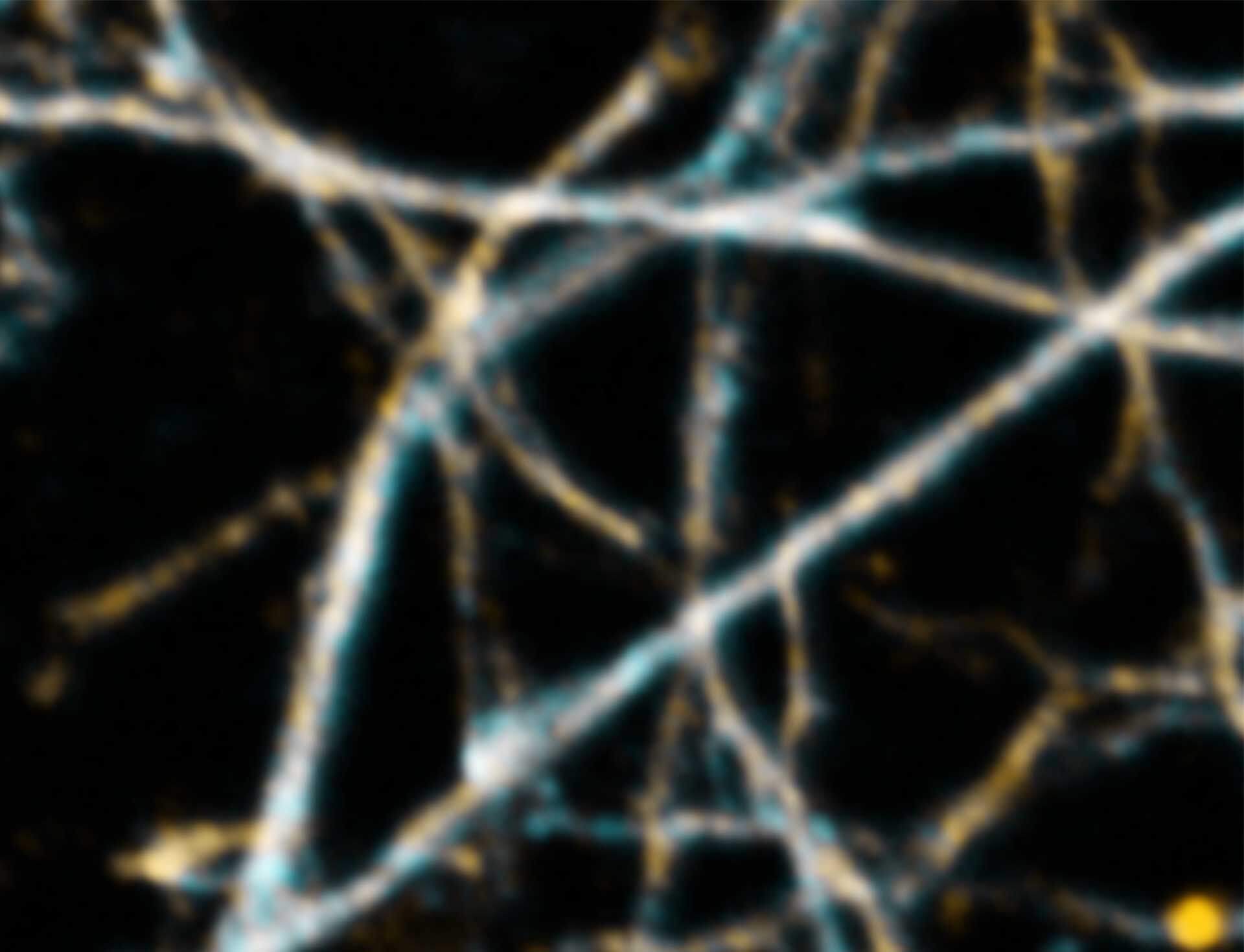
2D MINFLUX imaging of the cytoskeletal protein vimentin. Vimentin was labeled with Alexa Fluor 647 in fixed mammalian cells using indirect immunofluorescence. Note the individual filaments at intersections are invisible in the confocal image.
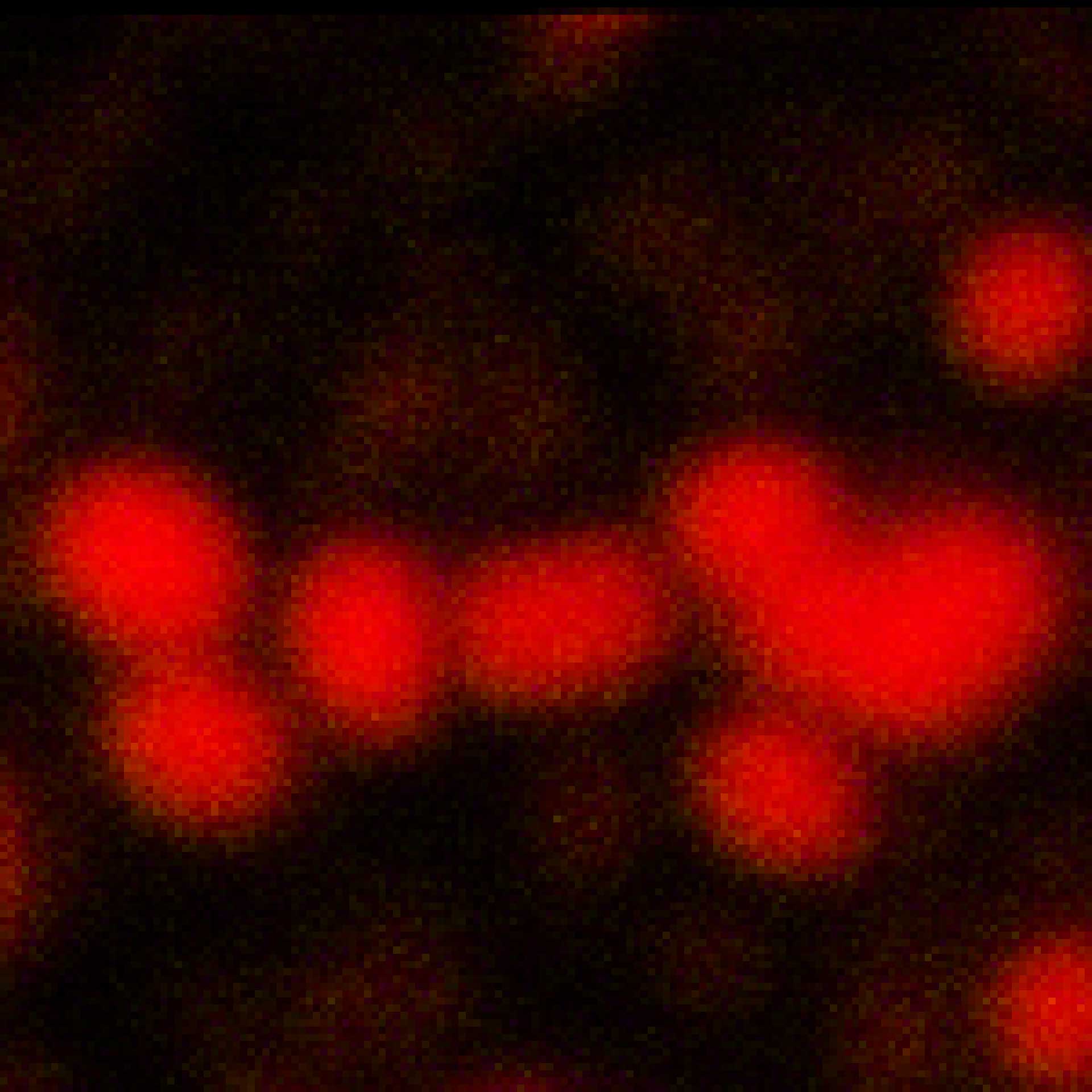

Description
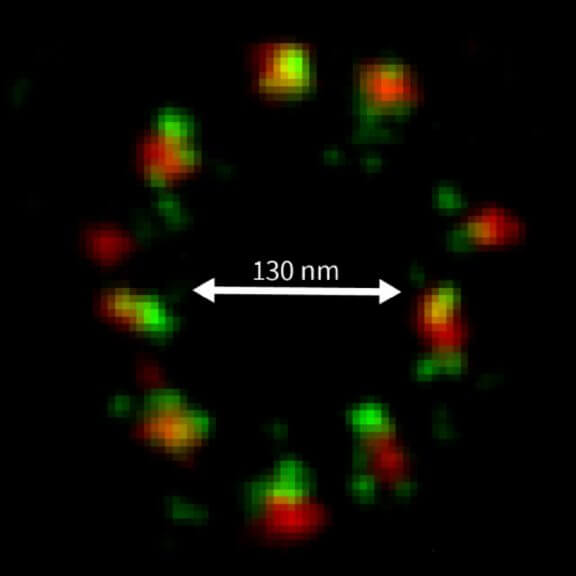
2D MINFLUX imaging of the peroxisomal membrane protein PMP70 labeled with Alexa Fluor 647 in fixed mammalian cells using indirect immunofluorescence

Description
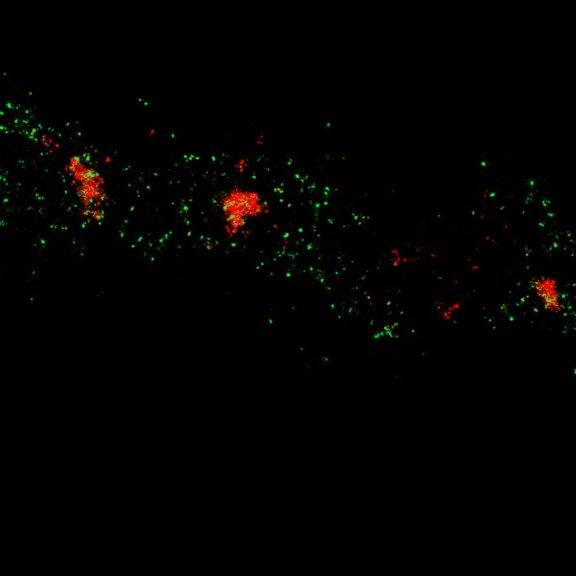
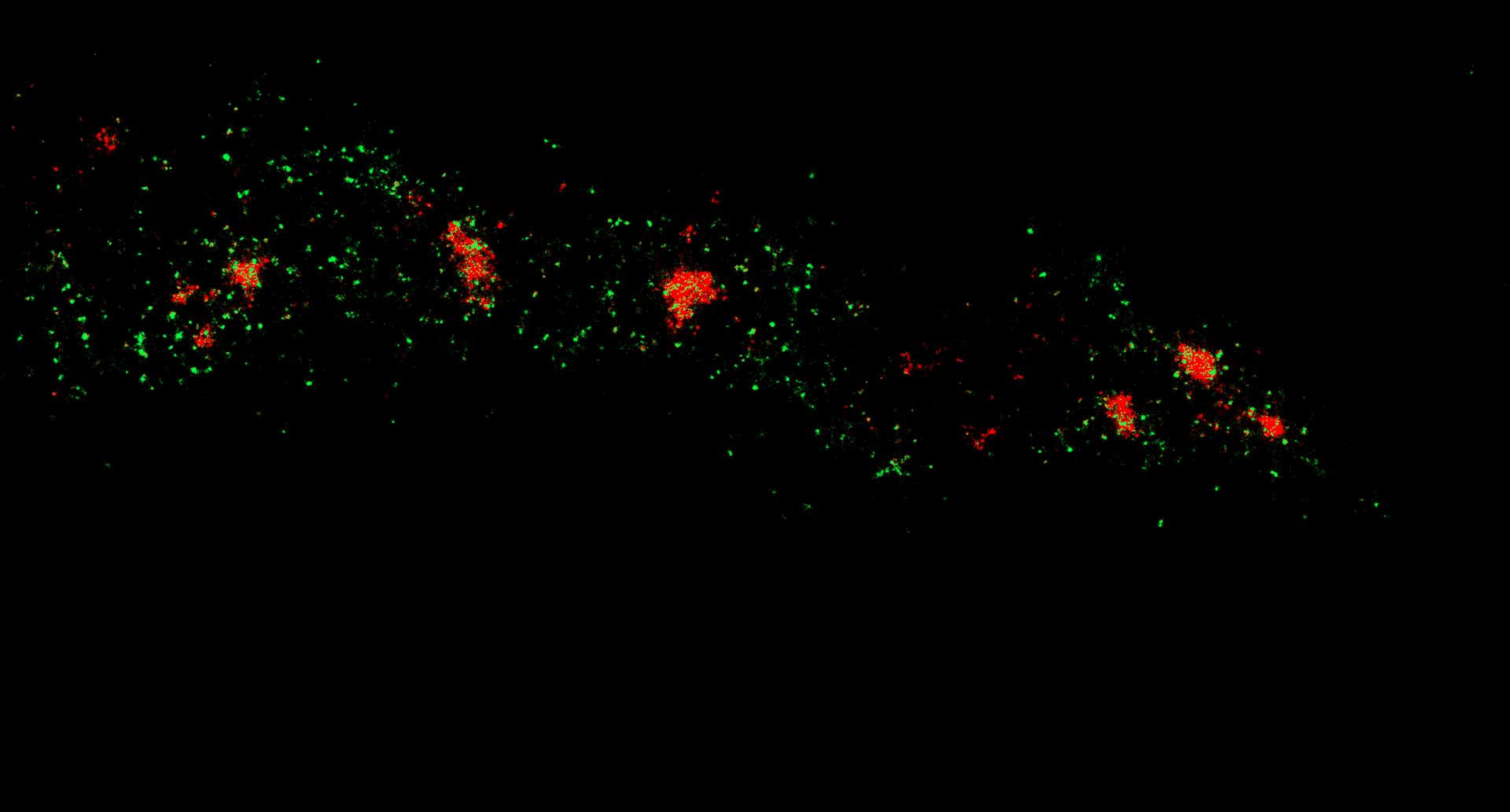
Two-color confocal and MINFLUX images of Tom20 (green) and mitochondrial DNA (red) stained with sCy5 and CF680 in mammalian cells using indirect immunolabeling. The two fluorophores were distinguished by ratiometric detection strategy. Note the dissimilar labeling density of the two imaged structures.

Description
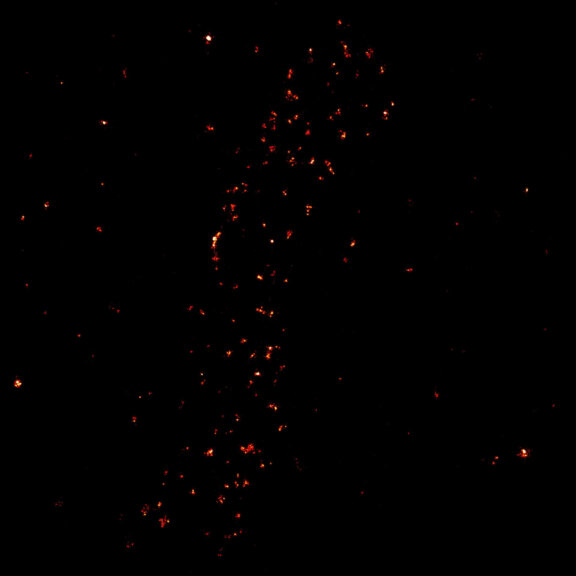
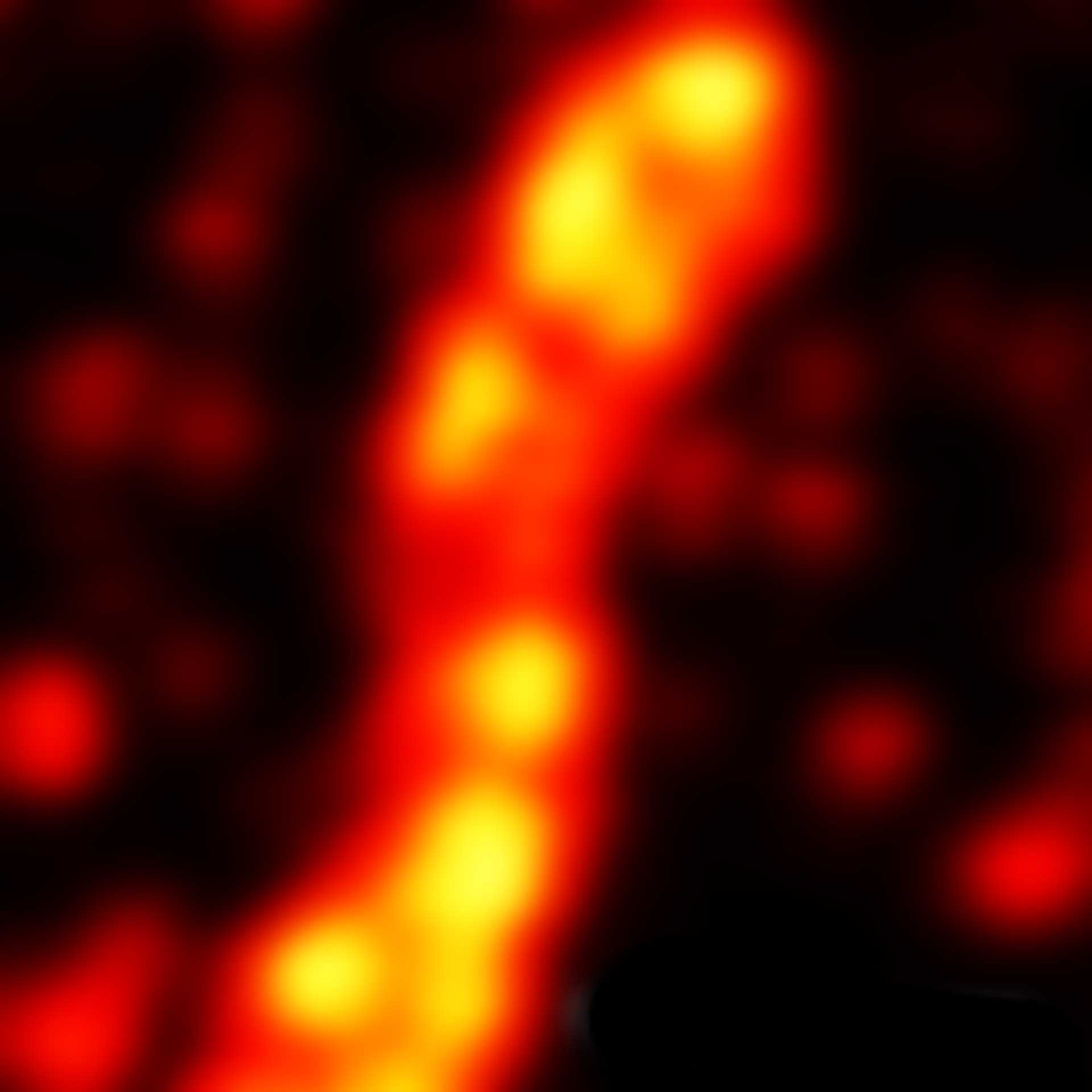
2D MINFLUX image of the mitochondrial import receptor Tom20 labeled with Alexa Fluor 647 in fixed mammalian cells using indirect immunolabeling.


Description
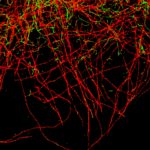
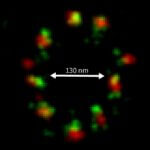
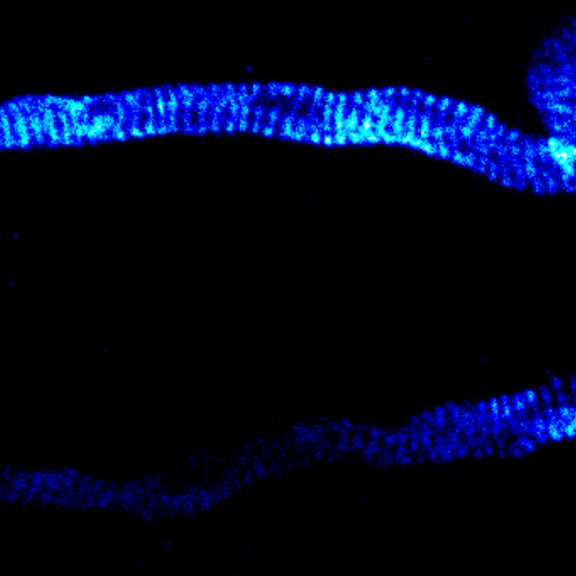
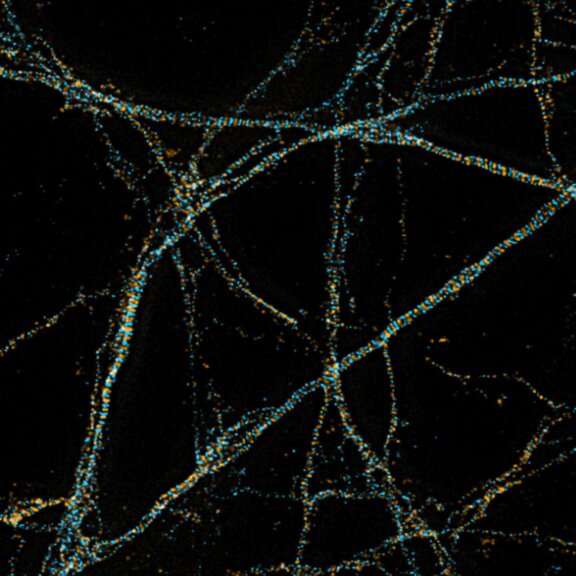
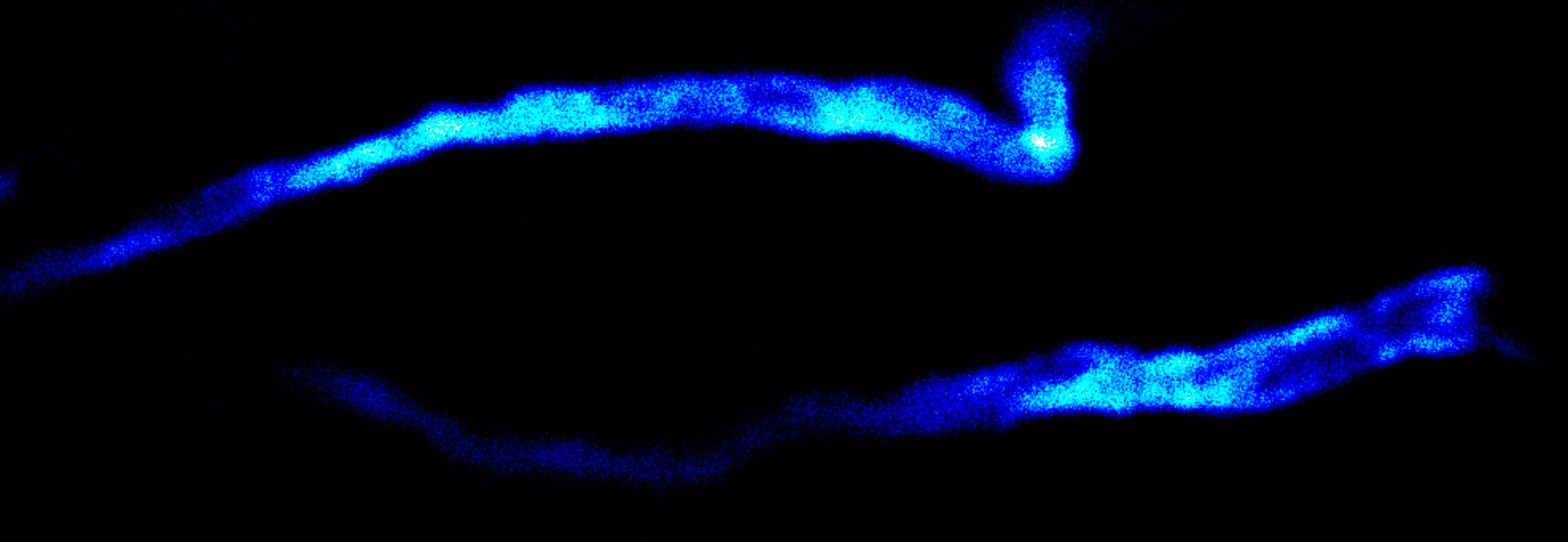
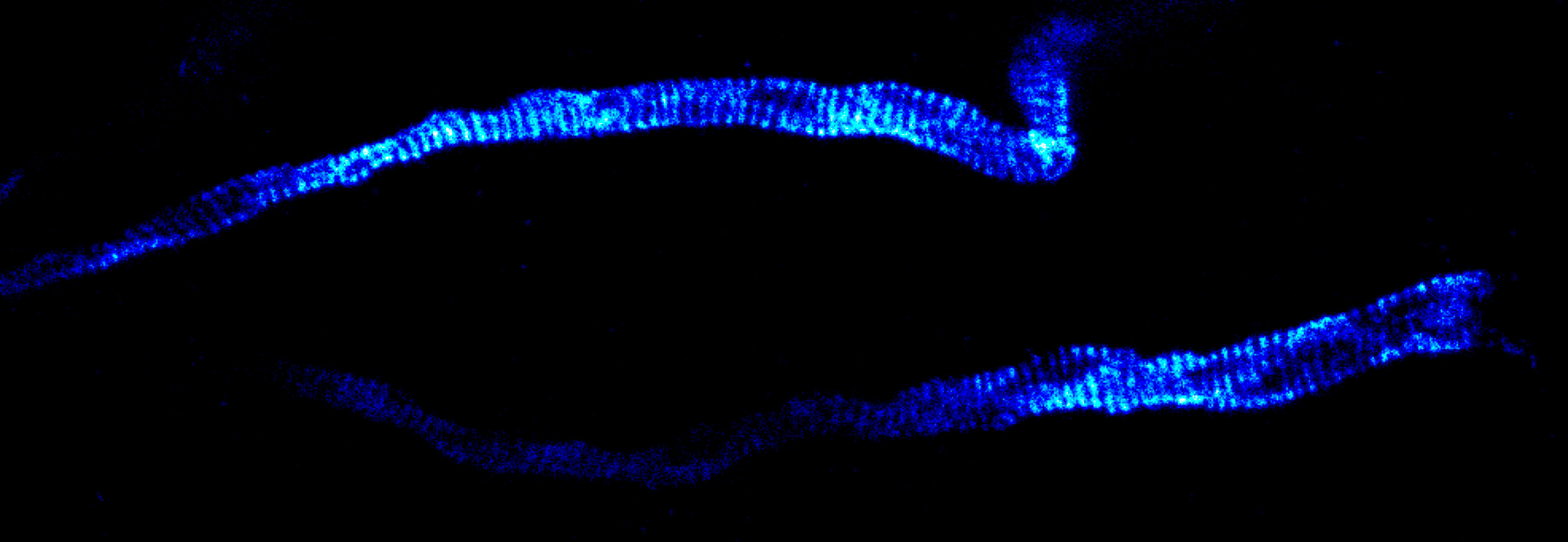
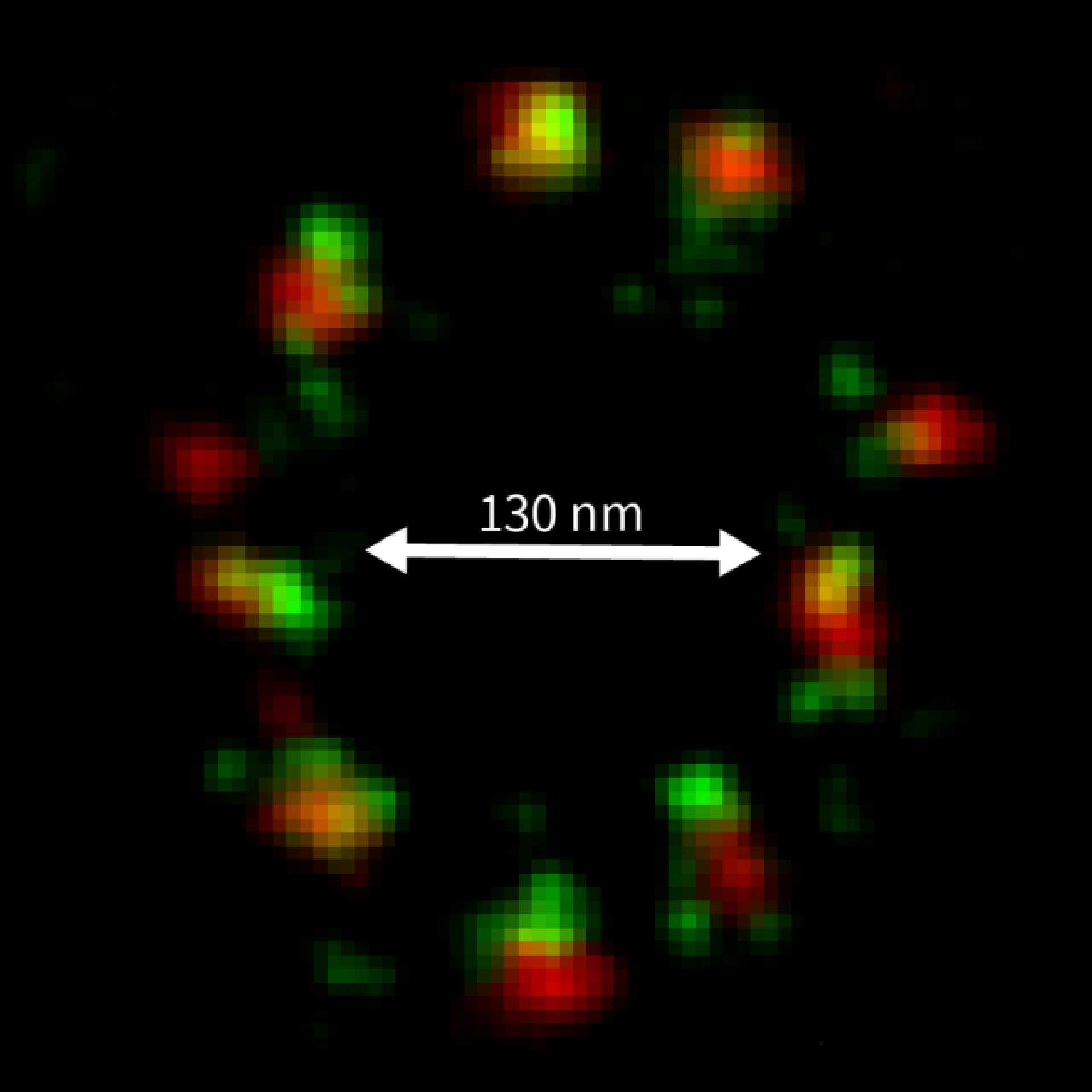
MINFLUX image of axonal bII spectrin in primary hippocampal neurons with < 2 nm resolution. Note the periodic arrangement of spectrin along the axon, and the absence of any details in the confocal counterpart image.
Description
Modules:

Description
Modules:
Description
Actin in microvilli of Caco2 cells. Fixed cells were stained with abberior STAR RED phalloidin. MATRIX detection removes background signal and dramatically improves optical sectioning for 2D-STED.
Samples by Prof. Dorothee Günzel & Roman Mannweiler, Institut für Klinische Physiologie, Charité - Universitätsmedizin Berlin.
Modules:
Description
Body wall preparation of a 3rd instar Drosophila melanogaster larva. Synapses at the neuromuscular junction are visualized by immunostaining against Bruchpilot (Brp), an integral component of active zones in Drosophila. Images were acquired on a STEDYCON attached to an Olympus IX81. Images were kindly provided by Dr. Nadine Ehmann, Institute of Physiology, Neurophysiology, University of Würzburg.
Description
Pollen autofluorescence imaged with a STEDYCON on an IX83 microscope frame. z-stacks on 95 tiles were acquired and stitched with SVI Huygens.
Description
Immunolabelling of βIV-spectrin, a scaffolding protein of the axon initial segment, using a self-made primary antibody and Alexa 594 secondary antibody. Cryo-section of mouse neocortex cut at 20 µm. Images were acquired using a STEDYCON attached to a Zeiss AxioImager Z2. The sample was kindly provided by Dr. Maren Engelhardt, Institute of Neuroanatomy, Medical Faculty Mannheim, Heidelberg University.
Description
Confocal imaging of a large sample region. Shown is a 2.8 mm x 2.5 mm region, acquired using 35 by 29 tiles using the STEADYFOCUS and stitched with SVI Huygens. Sample: mammalian cells immunolabelled for the mitochondrial protein TOM20 (abberior STAR RED, red), double-stranded DNA to visualize mitochondrial DNA (abberior STAR ORANGE, green), phalloidin to label F-actin (abberior STAR GREEN, gray), and DAPI to label nuclei. Sample preparation by abberior GmbH.
Description
TIMEBOW image of tubulin labeled with Atto647N and vimentin labeled with abberior STAR 635P. The two labels (Atto647N and abberior STAR 635P) have the same excitation spectra, but different lifetimes. Tubulin (red, short lifetime) and vimentin (green, long lifetime) can successfully be separated in the TIMEBOW STED recording via their lifetimes.
Modules:
Description
TIMEBOW image of mammalian cells labeled with antibodies against Tom20, ATTO 647N and NUP153, abberior STAR 635P. Note that nuclear pore (NUP) complex subunits appear to be localized in the cytoplasm, which is due to the NUP import pathway into the nuclear membrane.
Modules:
Description
TIMEBOW confocal and TIMEBOW STED image of mammalian cells labeled with antibodies against lamin, ATTO 647N and dsDNA, abberior STAR 635P.
Modules:
Description
2D tracking of a lipid-coupled Atto 647N molecule embedded in a supported lipid bilayer. The movement of the labeled lipid was tracked with a frequency of up to 10 kHz.
Description
2D MINFLUX imaging of the cytoskeletal protein vimentin. Vimentin was labeled with Alexa Fluor 647 in fixed mammalian cells using indirect immunofluorescence. Note the individual filaments at intersections are invisible in the confocal image.
Description
2D MINFLUX imaging of the peroxisomal membrane protein PMP70 labeled with Alexa Fluor 647 in fixed mammalian cells using indirect immunofluorescence
Description
Two-color confocal and MINFLUX images of Tom20 (green) and mitochondrial DNA (red) stained with sCy5 and CF680 in mammalian cells using indirect immunolabeling. The two fluorophores were distinguished by ratiometric detection strategy. Note the dissimilar labeling density of the two imaged structures.
Description
2D MINFLUX image of the mitochondrial import receptor Tom20 labeled with Alexa Fluor 647 in fixed mammalian cells using indirect immunolabeling.
Description
MINFLUX image of axonal bII spectrin in primary hippocampal neurons with < 2 nm resolution. Note the periodic arrangement of spectrin along the axon, and the absence of any details in the confocal counterpart image.