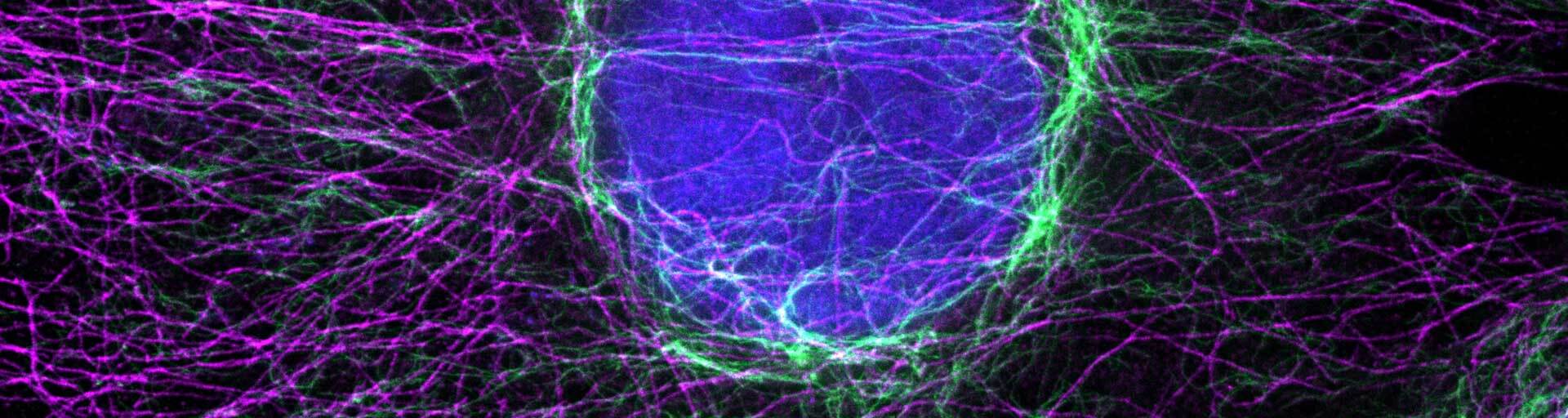
Immunolabeling with secondary nanobodies
The combination of super bright and highly photostable abberior dyes and secondary nanobodies offer a fantastic solution for high resolution imaging experiments where small labels, good penetration and outstanding specificity are necessary.
More information about:
Monoclonal FluoTag®-X2 sdABs, NanoTag Biotechnologies
FluoTag-X2® anti-mouse IgG1 abberior STAR 460L >
FluoTag-X2® anti-mouse IgG1 abberior STAR GREEN >
FluoTag-X2® anti-mouse IgG1 abberior STAR ORANGE >
FluoTag-X2® anti-mouse IgG1 abberior STAR RED >
FluoTag-X2® anti-rabbit IgG abberior STAR 460L >
FluoTag-X2® anti-rabbit IgG abberior STAR GREEN >
FluoTag-X2® anti-rabbit IgG abberior STAR ORANGE >
FluoTag-X2® anti-rabbit IgG abberior STAR RED >
Polyclonal Nanobodies, Jackson ImmunoResearch
abberior STAR RED nanobody anti-Mouse >
abberior STAR 580 nanobody anti-Mouse >
abberior STAR 460L nanobody anti-Mouse >
abberior STAR RED nanobody anti-Rabbit >
abberior STAR 580 nanobody anti-Rabbit >
abberior STAR 460L nanobody anti-Rabbit >