How much can you trust your labeling?
Highlights
- Dependable labeling is crucial for reliable fluorescence microscopy
- Critical control of labeling quality, antibody specificity, and expected localization prevents wrong conclusions and increases reviewer confidence
- Superresolution STED microscopy is ideal to evaluate labeling and sample quality by combining nanoscale resolution with multi-channel reference information
- Superresolution STED reveals labeling artifacts and phenotypes that are hidden in standard confocal imaging
Evaluating label localization accuracy beyond confocal limits
When a diffraction-limited image looks convincing
Fluorescence microscopy relies on the assumption that antibodies, nanobodies, fluorescent proteins, and drug conjugates accurately report the true localization of their target. Confocal microscopy is the standard for screening and routine imaging but fails for structures separated by less than ∼200 nm, creating diffraction-driven illusions and uncertainty. Is a labeled molecule located inside an organelle or associated to the outside of its membrane? Are two proteins truly colocalized or are their signals merely overlapping optically? Is a structure continuous, or composed of discrete nanoscale units?
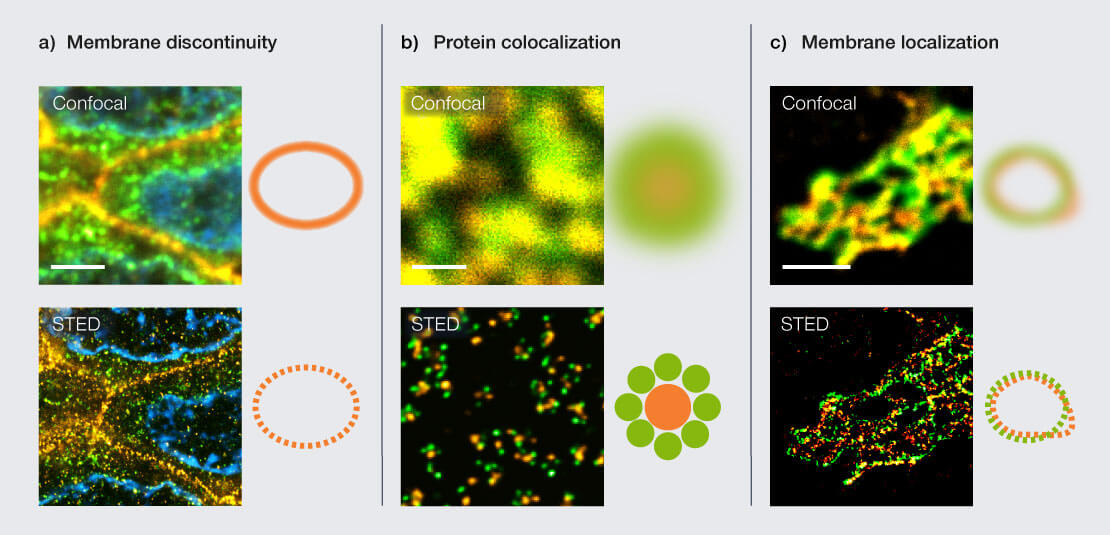
Figure 1: Scenarios for labeling control with STED microscopy. (a) Paraffin section of breast cancer tissue labeled for a nuclear pore protein (green, abberior STAR ORANGE), Her2 (orange, STAR RED), and Hoechst (cyan). Confocal imaging suggests a continuous membrane signal for HER2, while STED reveals discontinuous, punctate labeling, indicating suboptimal structural preservation or antibody binding. Scale bar 3 µm. (b) Nuclear pore complexes stained for two different nuclear pore proteins (green, STAR RED; orange, STAR ORANGE). Apparent colocalization in confocal microscopy resolves into distinct nanoscale protein distributions in STED. Scale bar 500 nm. (c) Golgi apparatus in mammalian cells labeled for the proteins GM130 (green, STAR RED) and giantin (orange, STAR ORANGE). High confocal colocalization disappears in STED, revealing distinct localization of proteins in relation to each other. Scale bar 2 µm.
Evaluating label quality with STED
Stimulated emission depletion (STED) physically overcomes the diffraction limit and resolves nano-scale organization down to ∼30 nm and below, providing direct structural evidence, not inferred resolution or artifact-prone computational reconstruction. It thus enables meaningful colocalization analysis, exposes unspecific binding and clustering artifacts, reveals nanoscale phenotypes, and produces robust data within a routine fluorescence workflow as it builds directly on confocal microscopy.
STED also improves overall image quality in multicolor images: using a single STED beam to deplete the signal from two or more different types of fluorophores guarantees that the two signals are perfectly coaligned and eliminates chromatic aberrations.
Reviewers increasingly expect superresolution evidence when claims about spatial organization at the nanoscale are made.
What STED reveals
Figure 1 shows three common scenarios where confocal microscopy suggests correct labeling, while STED reveals the true nanoscale organization: In paraffin tissue sections, confocal microscopy shows a smooth membrane signal, suggesting a continuous membrane structure. STED reveals that the signal consists of discrete nanoscale puncta (Fig. 1a). Confocal images suggest near-complete colocalization of nuclear pore proteins (Fig. 1b). Electron microscopy data, on the other hand, shows the overall ring-like structure of the nuclear pore but cannot to provide information about specific protein localization as options for protein labeling are severely limited in electron microscopy. Superresolution fluorescence microscopy like STED resolves distinct protein distributions in inner and outer rings, demonstrating that apparent overlap arises from diffraction rather than true spatial coincidence.
Confocal imaging shows strong colocalization of two membrane signals in the golgi apparatus, suggesting that both labeled proteins are localized in the same membrane or region of golgi (Fig. 1c). STED clearly separates the signals and reveals the distinct relative localization.
Knowing a label’s quality saves time and effort
Early control of label localization with STED saves time: one avoids spending hours imaging with poor labels. Labeling artifacts such as aggregation, sticky antibodies, or high background, are immediately visible with nanoscale resolution. You learn whether the fluorophore is localized in the right compartment or on the right membrane, and you gain direct cues on how to improve your protocol.
Most importantly, knowing a label’s quality prevents false conclusions drawn from misleading images that lead your research into the wrong direction.
From confocal to STED: easier than you think
Switching from confocal to STED is typically straightforward and can be done with your established sample preparation workflow, without complex biochemical protocols (like for expansion microscopy). In many cases, your standard confocal samples will work, and standard confocal imaging settings apply.
Conclusion
Confocal microscopy is a first step to assess label quality. Subsequently, superresolution helps you to better understand your sample and refine your research question.
STED microscopy visualizes labeling at the scale where biology is organized and may reveal otherwise invisible phenotypes. It saves time, prevents misleading conclusions, and strengthens reviewer confidence.
Authors
Martin Meschkat, Frederik Köpper